Session Information
Date: Saturday, November 12, 2022
Title: RA – Animal Models Poster
Session Type: Poster Session A
Session Time: 1:00PM-3:00PM
Background/Purpose: Inflammatory-erosive arthritis is exacerbated by lymphatic dysfunction (1). Mast cells (MCs) regulate lymphatic vessels by releasing inflammatory and vasoactive mediators (e.g. histamine) (2). Previous studies emphasized the pro-inflammatory role of MCs in rheumatoid arthritis (3), while recent studies identified their role in M2 vs. M1 macrophage transition, and anti-inflammatory effects in tissues (4). Here we aimed to elucidate the effects of MCs on lymphatic function and inflammatory-erosive arthritis in tumor necrosis factor transgenic (TNF-Tg) mice, which exhibit defects in ankle joint-draining popliteal lymphatic vessels (PLVs).
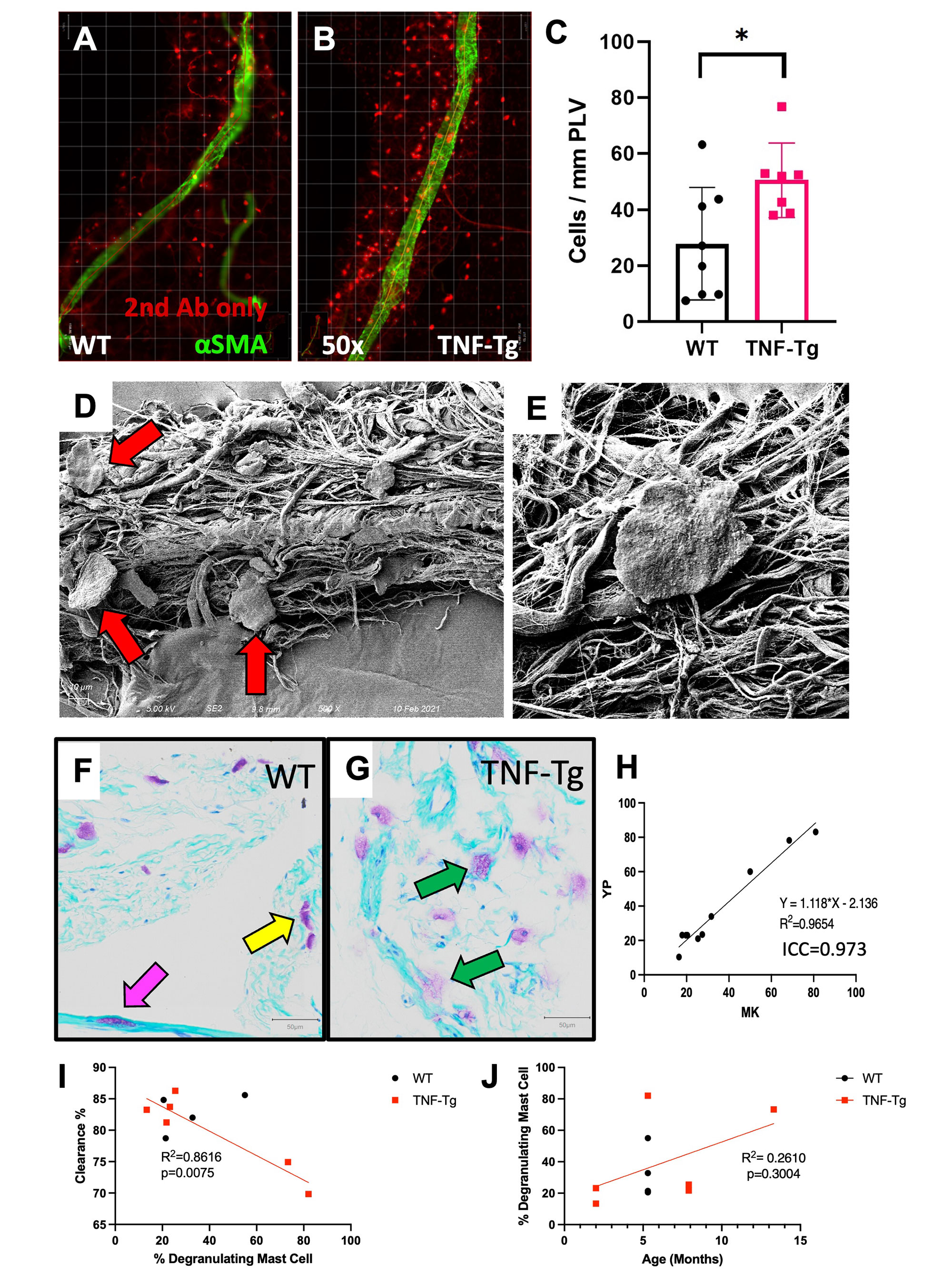
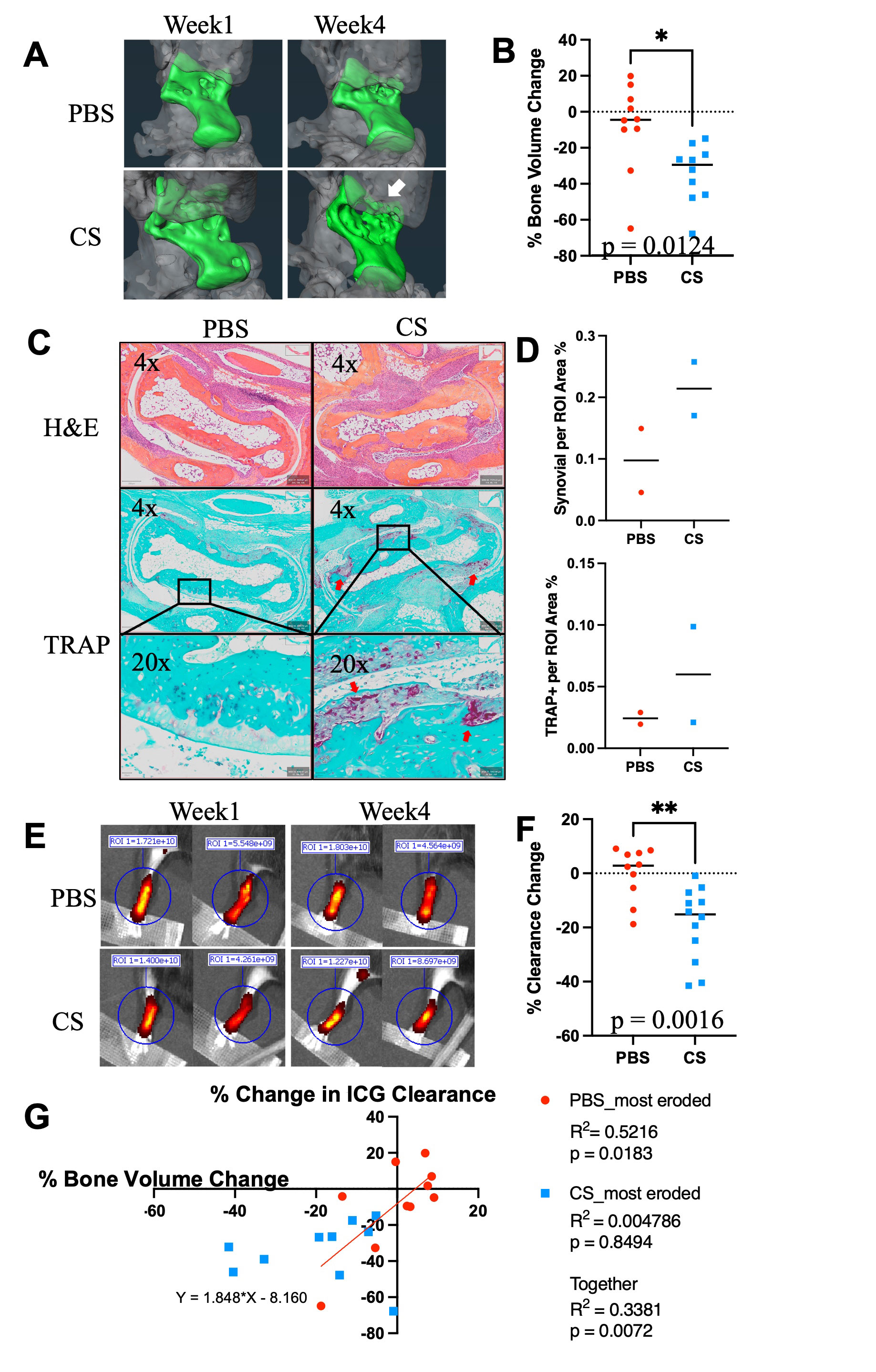
Methods: Whole mount immunofluorescent microscopy (WMIFM) and toluidine blue (TB) stained histochemistry were performed to quantify peri-PLV MCs. Scanning electron microscopy (SEM) was used to visualize peri-PLV MCs (n=2 wild-type (WT) mice). Ankle bone volumes were assessed by μCT as a biomarker of erosive arthritis in WT, TNF-Tg, and KitW-sh/W-sh (cKit-/-) mice (5) that have a selective hematopoietic deficit in MCs. Near-infrared indocyanine green (NIR-ICG) lymphatic imaging was used to quantify ICG clearance, an outcome measure of PLV draining function (1). 4-month-old female TNF-Tg mice received cromolyn sodium, a clinically used MC stabilizer (3.15mg/g/day/i.p., n=6), or saline (n=5) for 3 weeks. In vivo measurements including ICG clearance and μCT were collected at baseline and after 6-weeks of treatment, followed by ex vivo WMIFM and TB histology.
Results: WMIFM and TB histochemistry demonstrated a significant increase in the number of peri-PLV MCs in TNF-Tg vs. WT littermates, and SEM confirmed the peri-PLV localization of MCs (Figure 1). Moreover, the percentage of degranulating TB-stained MCs was inversely correlated with ICG clearance (Figure 1). TB-staining also identified intra-PLV MCs. Cromolyn sodium therapy significantly exacerbated TNF-induced bone loss, and decreased ICG clearance (Figure 2). Bone erosions and ICG clearance were also exacerbated in cKit-/- x TNF-Tg vs. TNF-Tg mice, which was associated with a similar reduction of PLV αSMA coverage in both groups (Figure 3).
Conclusion: TNF-Tg mice exhibit increased numbers of peri-PLV MCs with increased proportions of activated/degranulated MCs, which are inversely correlated with lymphatic function. Genetic ablation of MCs and pharmacological inhibition of MC degranulation both exacerbate TNF-induced inflammatory-erosive arthritis with decreased lymphatic clearance. Together, these findings support an inflammatory role of activated/degranulated peri-PLV MCs during arthritic progression, and a homeostatic role of intra-PLV MCs, in which loss of the later is dominant. Future studies to test this hypothesis towards elucidating MC regulation of lymphatic function and arthritic flare are warranted.
Reference
1. Bouta et al. Nat Rev Rheum. 14(2):94-106. 2018.
2. Pal et al. Front Immunol. 11:1234. 2020
3. Nigrovic et al. Arthritis Res Ther. 7(1):1-1. 2004
4. Wang et al. BioRxiv. 2021.
5. Grimbaldeston et al. Amer J Pathol. 167(3):835-48. 2005.
To cite this abstract in AMA style:
Peng Y, Kenney H, Bentley K, Xing L, Ritchlin C, Schwarz E. Protective Role for Mast Cells in TNF-induced Inflammatory-Erosive Arthritis and Its Associated Lymphatic Dysfunction in Mice [abstract]. Arthritis Rheumatol. 2022; 74 (suppl 9). https://acrabstracts.org/abstract/protective-role-for-mast-cells-in-tnf-induced-inflammatory-erosive-arthritis-and-its-associated-lymphatic-dysfunction-in-mice/. Accessed .« Back to ACR Convergence 2022
ACR Meeting Abstracts - https://acrabstracts.org/abstract/protective-role-for-mast-cells-in-tnf-induced-inflammatory-erosive-arthritis-and-its-associated-lymphatic-dysfunction-in-mice/