Session Information
Date: Sunday, November 8, 2020
Title: Epidemiology & Public Health III: Risk Factors & Outcomes (1472–1476)
Session Type: Abstract Session
Session Time: 5:00PM-5:50PM
Background/Purpose: Type 2 diabetes (T2D) and gout/hyperuricemia frequently coexist, but the nature and direction of this relationship is unclear. Observational studies have reported positive associations between gout/SU levels and risk of T2D but are subject to residual confounding or reverse causality bias. We used bidirectional two-sample Mendelian randomization to obtain unconfounded evidence of these potential causal effects.
Methods: Aggregate data from three large genome-wide association studies were used to identify genetic variants (SNPs) associated with SU levels (CKDGEN Consortium (Nat Genetics 2019; n=288,649 European-ancestry individuals; 123 SNPs)), T2D (DIAbetes Genetics Replication And Meta-analysis consortium (DIAGRAM: >34,000 T2D cases and >114,000 controls)), and related glycemic traits (Meta-Analyses of Glucose and Insulin-related traits Consortium (MAGIC): >200,000 in people without diabetes). These SNPs served as instrumental variables for the genetically determined exposures at conception, thus free of confounding by all factors after birth (e.g., lifestyle factors).
We first assessed the association between genetically-determined SU levels and T2D and levels of 3 glycemic traits, insulin resistance (HOMA-IR), fasting insulin (FI), and HbA1c, using inverse variance weighted (IVW) meta-analysis methods. We also separately examined the effects of SNPs from genes which regulate SU levels, SLC2A9 and ABCG2, estimating Wald ratios for these individual SNPs.
We then assessed the effects of genetically-determined T2D and glycemic traits on changes in SU levels, including and excluding a pleiotropic SNP (rs1260326) mapped to the GCKR gene.
In addition to the main IVW analyses, we generated MR-Egger, weighted median and mode, and MR-PRESSO estimates to account for potential heterogeneity and horizontal pleiotropy.
Analyses were performed with the TwoSampleMR and MR-PRESSO packages in R.
Results: Estimated effects of genetically-determined SU levels on each of the four outcomes (HOMA-IR, FI, HbA1c, and T2D) were small and non-significant (Table 1); this was consistent across MR estimates. The SLC2A9 and ABCG2 SNPs were strongly associated with SU levels (beta=0.33 mg/dL, R2=2.4% and beta=0.25 mg/dL, R2=0.7%, respectively; both p< 0.001), but neither were associated with T2D or any glycaemic traits (Table 1).
In the opposite direction, there was a strong association between genetically determined levels of FI and SU (beta=0.37 mg/dL, p=0.001) (Table 2), which strengthened after excluding the pleiotropic SNP in GCKR (beta=0.52 mg/dL (95% CI 0.35 to 0.66), p< 0.001).
Conclusion: Evidence from this Mendelian randomization analysis suggests that genetically raised fasting insulin, a measure of insulin resistance and precursor to T2D, is causally associated with hyperuricemia. Results are consistent with cross-sectional/observational studies and corroborate physiologic evidence for insulin’s anti-uricosuric property. Conversely, our data do not support a causal association of SU with FI levels or T2D risk. Interventions targeting SU levels are unlikely to lower T2D risk, whereas lowering insulin resistance would lower risk of gout.
 Table 1: Causal effect estimates for genetically-determined serum urate levels (per 1 mg/dL) on type 2 diabetes risk and other glycemic trait levels
Table 1: Causal effect estimates for genetically-determined serum urate levels (per 1 mg/dL) on type 2 diabetes risk and other glycemic trait levels
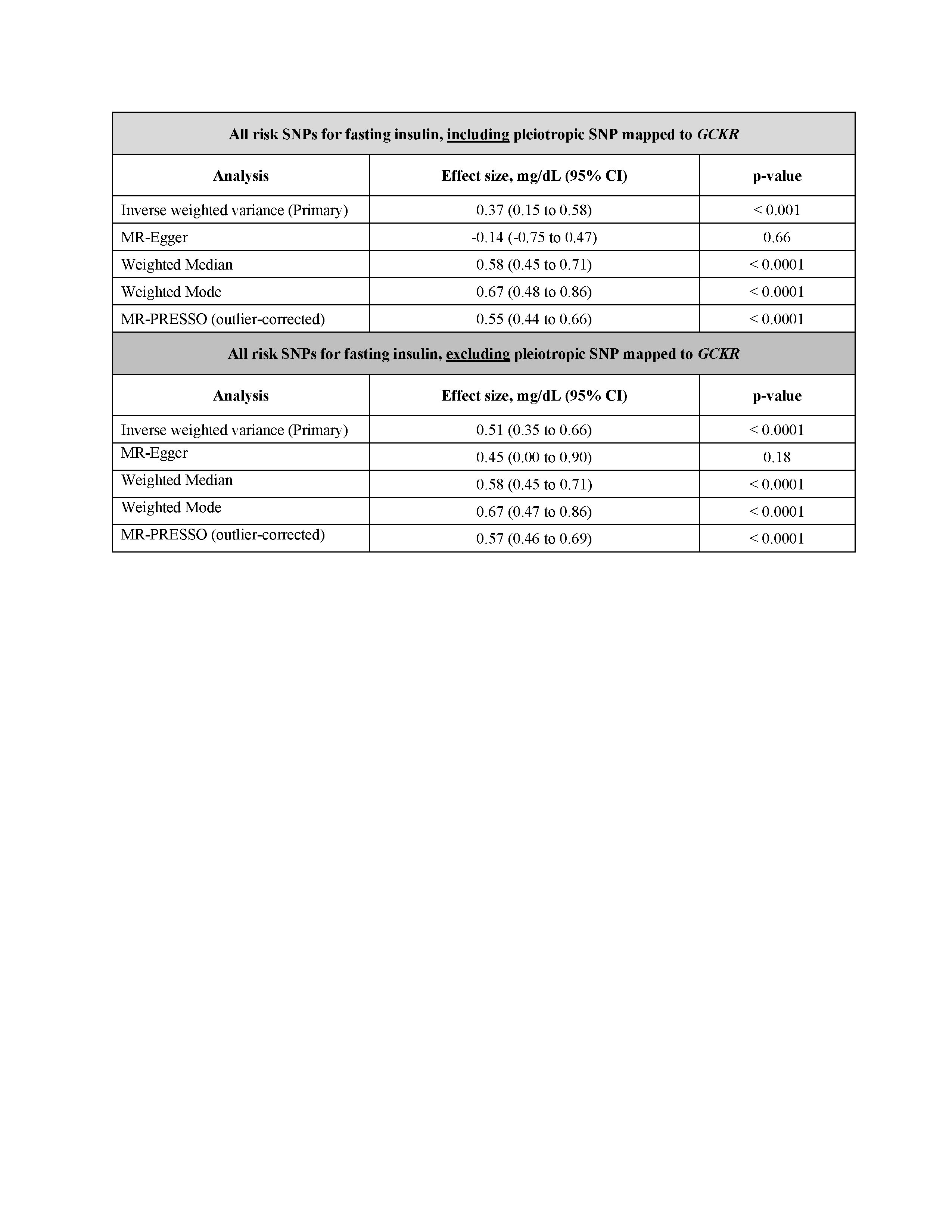 Table 2: Causal effect estimates for genetically-determined levels of fasting insulin (ascertained in non-diabetics) on levels of serum urate (mg/dL)
Table 2: Causal effect estimates for genetically-determined levels of fasting insulin (ascertained in non-diabetics) on levels of serum urate (mg/dL)
To cite this abstract in AMA style:
McCormick N, O'Connor M, Marozoff S, Choi J, Leong A, Choi H. Assessing Causal Associations of Urate Levels with Type 2 Diabetes and Related Glycemic Traits Using Bidirectional Mendelian Randomization [abstract]. Arthritis Rheumatol. 2020; 72 (suppl 10). https://acrabstracts.org/abstract/assessing-causal-associations-of-urate-levels-with-type-2-diabetes-and-related-glycemic-traits-using-bidirectional-mendelian-randomization/. Accessed .« Back to ACR Convergence 2020
ACR Meeting Abstracts - https://acrabstracts.org/abstract/assessing-causal-associations-of-urate-levels-with-type-2-diabetes-and-related-glycemic-traits-using-bidirectional-mendelian-randomization/
