Session Information
Session Type: ACR Poster Session A
Session Time: 9:00AM-11:00AM
Background/Purpose: Recent studies identify gut microbiota dysbiosis as a possible contributor to rheumatoid arthritis (RA) pathogenesis. RA patients have significantly different microbiomes than healthy controls. The K/BxN mouse model of arthritis does not develop disease in germ free conditions but adding commensal bacterial triggers arthritis in these mice. Sexual dimorphism may also contribute to differences in RA incidence, severity, and mortality. We recently described sex differences in the tumor necrosis factor transgenic (TNF-Tg) mouse model of RA wherein females have a decreased lifespan (166 vs 229 days). Here, we initiated a study to elucidate the microbiomes of male and female TNF-Tg mice.
Methods: Fecal samples were collected from 5.5 month male and female TNF-Tg mice that were housed separately (C57BL/6 background, n=10), an age when females have exacerbated disease. 16s DNA sequencing was performed to identify bacterial diversity. Beta-diversity and principal coordinate analysis (PCoA), i.e. a 3D representation of the specific differences between mice, were used to estimate bacterial diversity. Least discriminant analysis (LDA) was applied to determine significantly enriched (LDA score>2) species between sexes.
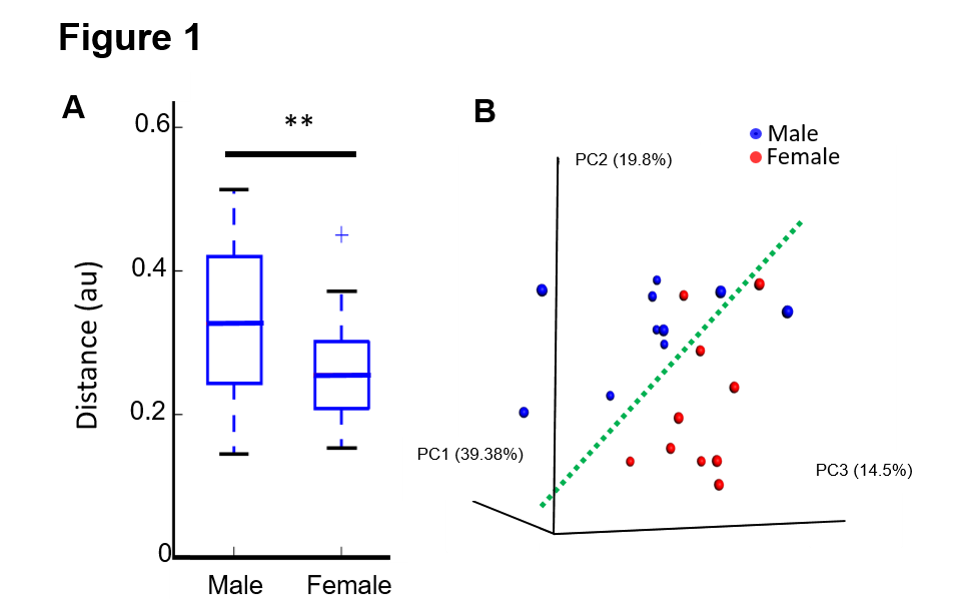
Results: Females have significantly decreased beta-diversity (p<0.05), and PCoA demonstrated that females have a distinctly different microbiome based on a clear spatial separation (Fig. 1 A and B). LDA determined that females were significantly enriched in Bacteroidetes and males in Firmicutes (Fig. 2).
Conclusion: Dysbiotic microbiota, loss of bacterial diversity, and sex differences separately have been associated with several autoimmune and inflammatory diseases. Here we show for the first time that female TNF-Tg mice with advanced arthritis have significantly less microbial diversity than males. Furthermore, the female microbiome differs in part due to enrichment of the pathobiont Bacteroidetes whereas males display enrichment of Firmicutes. Others have reported that an altered ratio of Bacteroidetes/Firmicutes influences autoimmune and inflammatory disease progression. This initial study assessed sexually dimorphic microbiota in TNF-Tg mice, and analyses of wild-type littermates are underway. Since the published microbiomes of male and female C57BL/6 mice do not differ in beta-diversity or specific species enrichment, we hypothesize that the loss of diversity and enrichment in Bacteroidetes significantly contributes to the disease progression in female TNF-Tg mice.
To cite this abstract in AMA style:
Bell R, Wood R, Ritchlin CT, Schwarz E, Rahimi H. Sexually Dimorphic Dysbiosis of Gut Microbiota in Tumor Necrosis Factor Transgenic Mice with Inflammatory-Erosive Arthritis [abstract]. Arthritis Rheumatol. 2016; 68 (suppl 10). https://acrabstracts.org/abstract/sexually-dimorphic-dysbiosis-of-gut-microbiota-in-tumor-necrosis-factor-transgenic-mice-with-inflammatory-erosive-arthritis/. Accessed .« Back to 2016 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/sexually-dimorphic-dysbiosis-of-gut-microbiota-in-tumor-necrosis-factor-transgenic-mice-with-inflammatory-erosive-arthritis/