Session Information
Session Type: Poster Session (Monday)
Session Time: 9:00AM-11:00AM
Background/Purpose: SLE is a chronic autoimmune disorder in which periods of relative disease inactivity are punctuated by flares that present with increased inflammation and tissue damage. The current literature suggests that this enhanced disease activity is linked to immune cell subsets involved in ANA production, including T follicular (Tfh) and peripheral helper (Tph) cells, plasma cells, and age-associated B cells (ABCs); however, it is largely unknown how these and other immune subsets differ between flare and inactive disease, particularly in individual patients followed longitudinally.
Methods: 12 healthy controls (HC), 23 SLE patients with flare (Active) and 9 inactive SLE patients (Inactive) were recruited. All patients had a diagnosis of SLE based on the 1997 ACR criteria. Active patients had to be within 1 month of flare onset (defined as a clinical SLEDAI-2K≥0 requiring a change in therapy) and agree to follow-up assessments at 6 and 12 months. Inactive patients had to be inactive for ≥1 year (clinical SLEDAI-2K=0) and be on ≤10mg prednisone at recruitment. Peripheral blood mononuclear cells (PBMCs) were isolated over a Ficoll gradient within 3 hrs of collection, stained with fluorescent antibodies for immune cell subsets, and examined by flow cytometry. Statistical significance was determined using Kruskal-Wallis tests to compare HC, active and inactive SLE groups, Wilcoxon matched-pairs signed rank tests to compare baseline and follow-up visits, and Spearman tests for correlation.
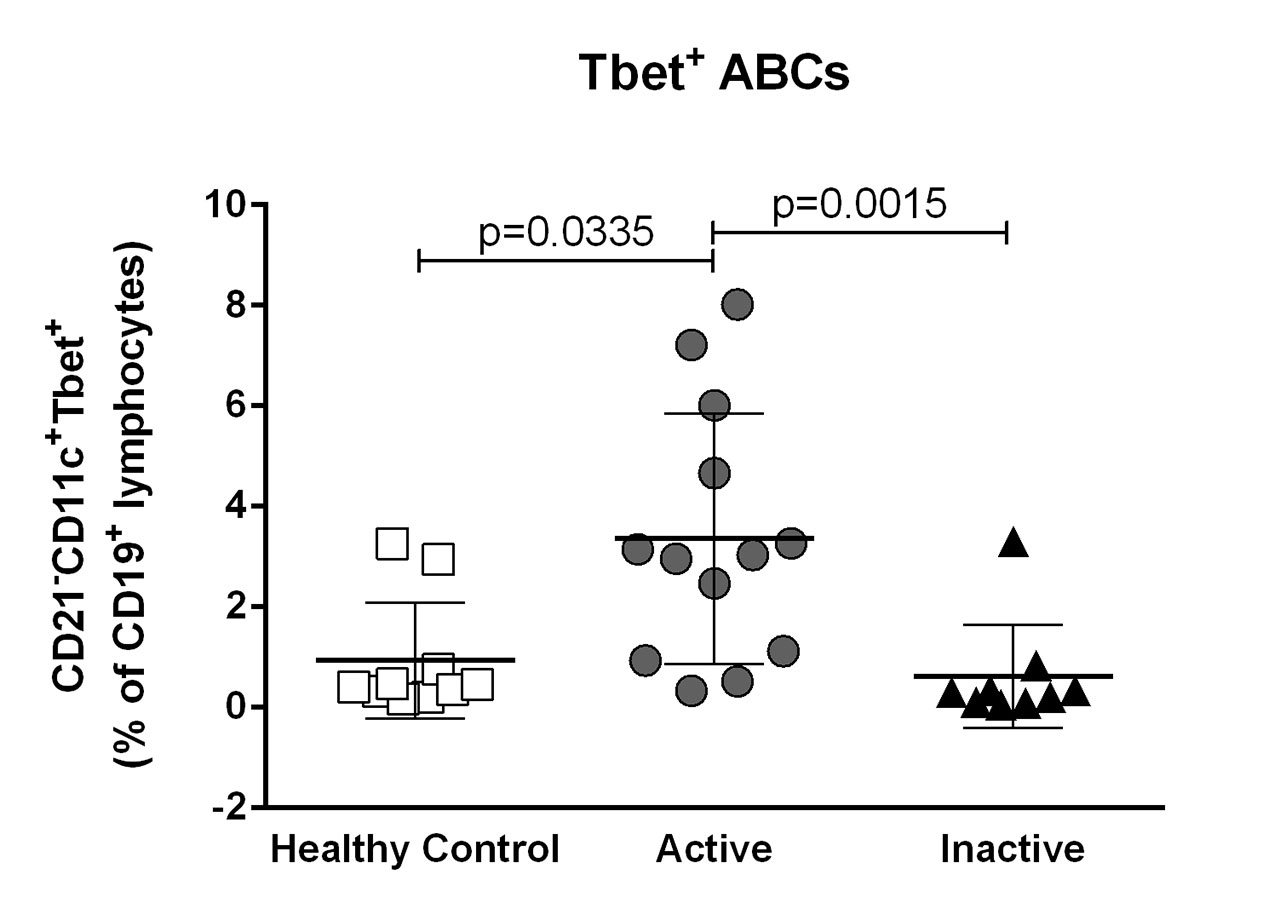
Results: While active and inactive patients had increased proportions of Tph cells (CD4+PD1hiCXCR5–) compared to HC, only a small subset of patients in either group had elevated levels of plasma cells (CD19loCD27hiCD38+CD138+), plasmablasts (CD19loCD27hiCD38+CD138–) and activated memory Tfh cells (CD4+PD1hiCXCR5hi), with no differences between the two groups. In contrast, active patients had significantly more ABCs (CD19+CD21–CD11c+Tbet+) than inactive patients or HC and this correlated significantly with Tph cells (r=0.6909, p=0.0226). Interestingly, at their 6-month follow-up visit, active patients showed significant increases in Tfh and plasma cells, and trends to increased CD86+ B and Tph cells, despite improvements in their SLEDAI-2K. These findings suggest that activated T and B cells may be depleted from the blood during flares, possibly as a result of tissue recruitment. Notably, increases in activated memory Tfh cells post-flare were predominantly due to increases in the Tfh17 subset, while the Tfh2 subset was significantly decreased.
Conclusion: Our results show that while Tph cells and ABCs are elevated in active SLE, plasma cells and activated Tfh cells may be depleted from the peripheral blood during acute flare and return to the blood post-flare, reflecting recruitment to and subsequent release from tissues. These findings highlight the importance of longitudinal studies in individuals with active SLE.
To cite this abstract in AMA style:
Manion K, Touma Z, Bonilla D, Gladman D, Urowitz M, Wither J. Dynamic Changes During SLE Flare Implicate Age-Associated B Cells and Altered T Follicular and Peripheral Helper Cell Responses in Disease Activity [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/dynamic-changes-during-sle-flare-implicate-age-associated-b-cells-and-altered-t-follicular-and-peripheral-helper-cell-responses-in-disease-activity/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/dynamic-changes-during-sle-flare-implicate-age-associated-b-cells-and-altered-t-follicular-and-peripheral-helper-cell-responses-in-disease-activity/