Session Information
Session Type: Abstract Session
Session Time: 11:00AM-12:30PM
Background/Purpose: Neutrophils are main mediators of disease in ANCA associated vasculitis (AAV). Pathogenic ANCA IgG on neutrophils promotes neutrophil adhesion and migration at the endothelium. NETs are present in patients with active AAV (aAAV), and vascular inflammation and occlusion occurs as a result. Sites of inflammation and tissue injury are presumably hypoxic in AAV, and whether pathological hypoxia enhances these effects remains unknown.
Methods: Neutrophils were isolated from healthy controls (HC) and patients with aAAV using immunomagnetic separation. For TEM and protein secretion, glomerular endothelial cells (gEC) were cocultured with neutrophils from HC for 1.5h to assess neutrophil TEM towards chemoattractant (IL-8, 200 pg/mL) and protein secretion (without IL-8 treatment) at 21% O2 or 1% O2. Isolated neutrophils were primed (TNF) and stimulated with MPO- or PR3-ANCA IgG or control IgG (CIgG) in 21% or 1% O2. Transmigrated neutrophils were counted via haemocytometer. Protein secretion was measured in supernatant by ELISA. NETosis was visualised by staining for H3Cit in stimulated neutrophils. For RNA-Sequencing (RNA-Seq), cells were lysed, RNA was extracted, and samples were sequenced using poly(A) RNA-Sequencing.
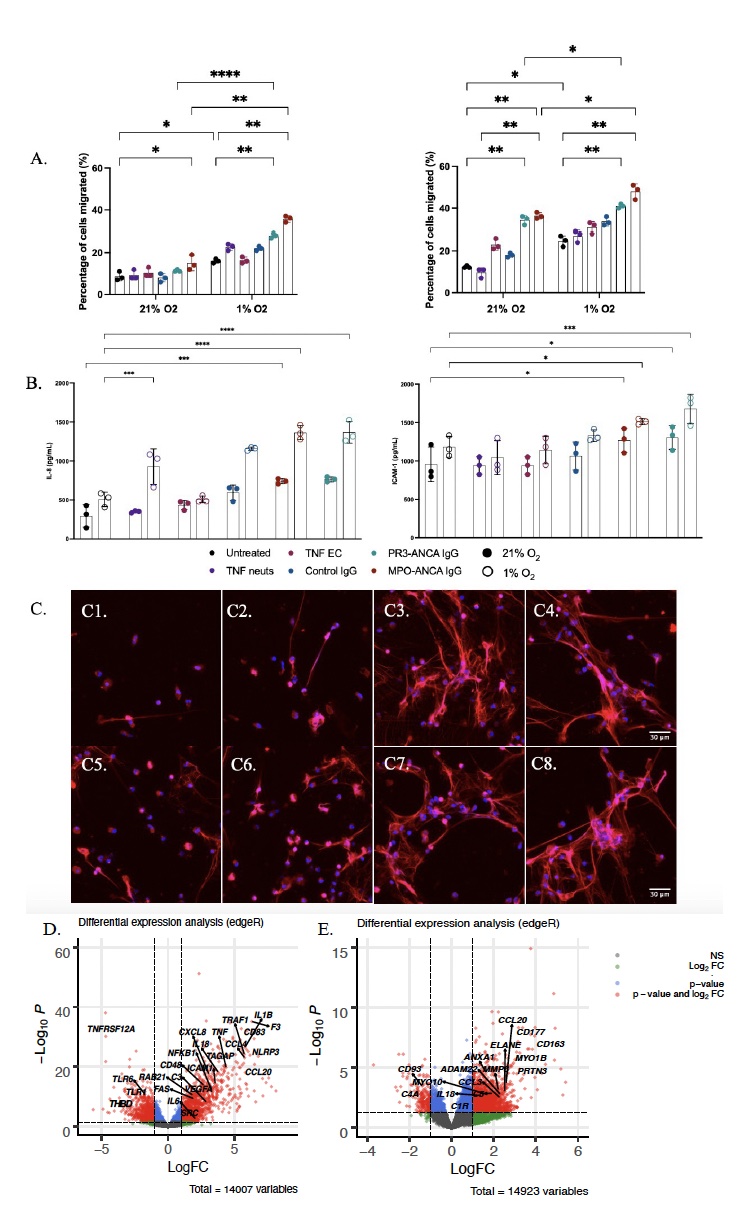
Results: At 21% O2, TEM across a gEC monolayer towards chemoattractant (IL-8) increased following MPO- and PR3-ANCA stimulation compared to untreated neutrophils (Fig1A; MPO: p=0.0017, PR3: p=0.0021). At 1% O2, there was a significant increase in neutrophil TEM in all conditions in comparison to 21% O2. Increased IL-8 secretion was seen in MPO- and PR3-ANCA stimulated coculture at 21% O2 (MPO: p=0.0004, PR3: p=0.0003). This was enhanced by incubation at 1% O2 (MPO: p=0.0003, PR3: p=0.0001) (Fig1B). Similar results were seen for ICAM-1 secretion (Fig1B). MPO- and PR3-ANCA stimulation increased NET formation at 21% O2 in comparison to untreated and CIgG stimulated neutrophils and this effect was enhanced by incubation at 1% O2 (Fig 1C). RNA-Seq identified IL-8 (CXCL8), ICAM-1, and Tissue Factor (F3) being significantly upregulated and thrombomodulin (THBD) being significantly downregulated differentially expressed (DE) genes (p< 0.05, Log2FC < 0.05) in MPO- and PR3-ANCA stimulated neutrophils at 21% and 1% O2 in comparison to untreated cells. CD177 and PRTN3 were significantly upregulated DE genes in isolated neutrophils from patients with aAAV in comparison to HC.
Conclusion: In comparison to untreated neutrophils, isolated HC neutrophils stimulated with ANCA undergo increased TEM across a gEC monolayer, increased protein secretion (IL-8 and ICAM-1) in neutrophil-gEC co-culture, increased NET formation, and upregulation of gene expression of IL-8, ICAM-1, and F3. This pathogenic effect of ANCA shown at 21% O2 in vitro is enhanced when neutrophils are incubated in vitro at 1% O2. Increased adhesion factors are present in the renal glomeruli, and increased neutrophil CD177 and PRTN3 expression in aAAV facilitates TEM through interaction with PECAM-1. The effect of ANCA and hypoxia regulated increased TEM in HC neutrophils may therefore be more robust in aAAV. In aAAV, hypoxia may enhance ANCA promoted neutrophil induced endothelial injury.
A. Neutrophil gEC co-culture TEM at 21% and 1% O2 without and with IL-8 (n=3) at 1.5h.
B. Neutrophil gEC co-culture protein (IL-8 and ICAM_1) secretion via ELISA at 21% and 1% O2 (n=3) at 1.5h.
C. NET (H3Cit, magenta) formation in neutrophils (DAPI, blue) at 21% and 1% O2 at 4h (n=5), 40x. 1/5: Untreated; 2/6: CIgG; 3/7: MPO-ANCA, 4/8: PR3-ANCA.
D. Differential expression analysis 21% and 1% O2 untreated v MPO-ANCA v PR3-ANCA (n=6).
E. Differential expression analysis untreated age match HC (n=10) v aAAV (n=20).
To cite this abstract in AMA style:
Pisacano N, Dhutia A, Rothery S, Pusey C, Chilvers E, Cowburn A, Lodge K, Prendecki M. ANCA IgG Promotion of Neutrophil Recruitment, Migration and Vascular Damage in ANCA Associated Vasculitis May Be Enhanced by Hypoxia [abstract]. Arthritis Rheumatol. 2024; 76 (suppl 9). https://acrabstracts.org/abstract/anca-igg-promotion-of-neutrophil-recruitment-migration-and-vascular-damage-in-anca-associated-vasculitis-may-be-enhanced-by-hypoxia/. Accessed .« Back to ACR Convergence 2024
ACR Meeting Abstracts - https://acrabstracts.org/abstract/anca-igg-promotion-of-neutrophil-recruitment-migration-and-vascular-damage-in-anca-associated-vasculitis-may-be-enhanced-by-hypoxia/