Session Information
Date: Monday, November 9, 2020
Title: SLE – Diagnosis, Manifestations, & Outcomes Poster III: Bench to Bedside
Session Type: Poster Session D
Session Time: 9:00AM-11:00AM
Background/Purpose: Systemic lupus erythematosus (SLE) is a chronic autoimmune disease driven by complex immune dysregulation, involving altered immune mediators and accumulation of autoantibody (AutoAb) specificities. We have previously identified patterns of disordered immunity associated with development of SLE (Munroe et al. Ann Rheum Dis. 2016; Lu et al. J. Autoimmun. 2016). This study seeks to determine an optimal panel of markers that can distinguish those SLE patients with active disease and help to refine a Lupus Disease Activity Immune Index (LDAII).
Methods: We procured 200 samples from 150 patients classified with SLE on dates of low disease scores measured by the hybrid SLEDAI (hSLEDAI) (< 4, range 0-3, n=100) or active disease (≥ 4, range 4-30, n=100). Race/sex/age-matched healthy control (Ctl) samples (n=50) were also evaluated. SLE-linked plasma immune mediators (n = 35) were evaluated by EllaTM microfluidic immunoassay and serum SLE-associated AutoAb specificities, including dsDNA, chromatin, Ro/SSA, La/SSB, Sm, SmRNP, RNP, and ribosomal P, were assessed by Bioplex® 2200 xMAP assay. A subset of 24 log-transformed immune mediators were further evaluated using random forest applied machine learning modeling to determine an optimal subset of mediators to inform the LDAII. The LDAII is the sum of log-transformed, standardized immune mediators, weighted by the Spearman r correlation coefficient of immune mediator levels vs. either hSLEDAI scores or number of SLE-associated AutoAb specificities, which correlates with clinical disease activity (Spearman r = 0.289, p< 0.0001).
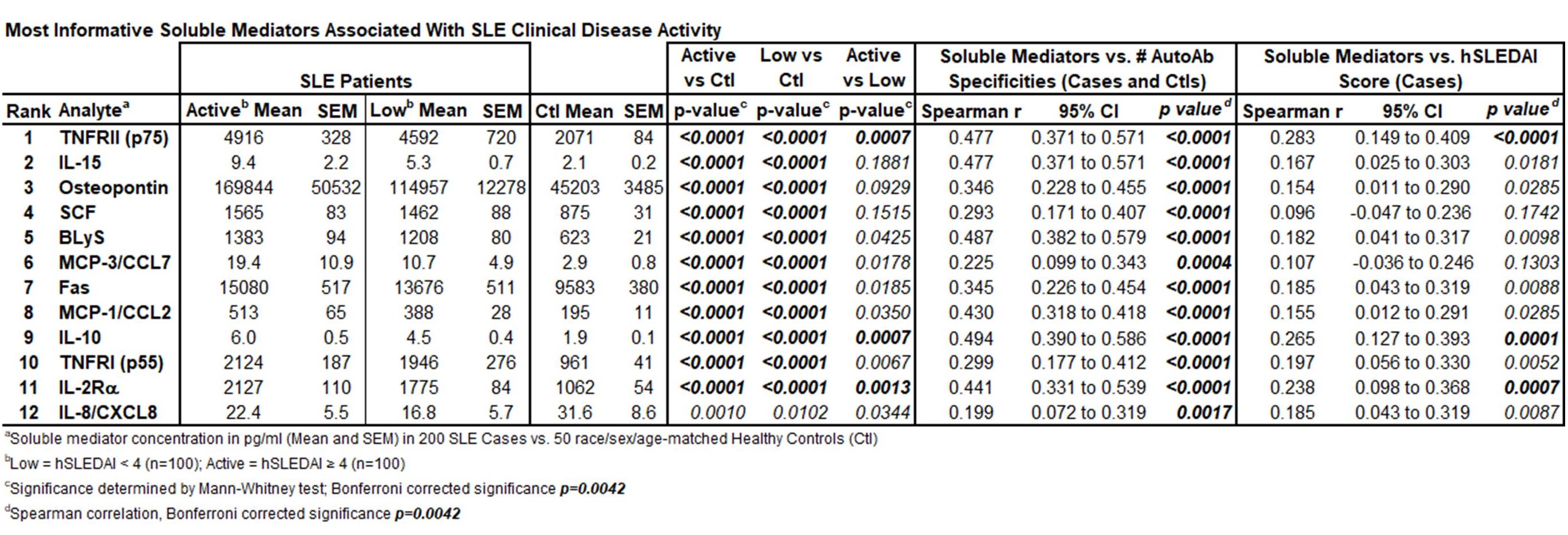
Results: No difference in age, ethnicity, or sex was noted between low or active clinical disease. After adjusting for multiple comparisons (Bonferroni corrected p< 0.0021), IFN-α, IL-2Rα, TNF-α, TNFRII, IP-10, MIG, and IL-10 remained significantly correlated with hSLEDAI scores (Spearman r=0.221-0.388), and 22/35 soluble mediators significantly correlated with accrued SLE-associated autoantibodies, including the top 12 LDAII informative mediators listed in the Table. Forward selection of the 24 mediators ranked by variable importance (Figure [A]) drew 12 mediators that best distinguished between controls, low disease and active disease (Table). These top 12 immune mediators best informed and maximized the performance of a newly refined LDAII (Figure [B]). The LDAII, weighted either by hSLEDAI score (p< 0.0001) or number of AutoAb specificities (p=0.0060), significantly correlated with disease activity in SLE patients and identified patients with renal involvement (p< 0.01), where SCF and TNFRII (p< 0.0001) were significantly altered. In addition, the LDAII distinguished patients with dsDNA binding and low complement as well as clinically and serologically active vs. quiescent disease states (p< 0.0001), differentiated by TNF-α, IL-2Rα, IP-10, and IL-10 levels (p≤0.001)
Conclusion: We have refined the LDAII in order to characterize SLE patients with active clinical disease. Treatment to target using a sensitive and objective biologic surrogate for clinical disease activity could help improve disease management and prevent organ damage in SLE.
To cite this abstract in AMA style:
Munroe M, DeJager W, Macwana S, Tran L, Guthridge J, Jupe E, DeFreese D, Newhardt R, Purushothaman M, Sharma S, Redinger N, Aberle T, Kamp S, Arriens C, Chakravarty E, Merrill J, James J. Ability of Innate, Adaptive, and TNF-Superfamily Immune Pathways to Characterize Disease Activity and Inform a Refined Lupus Disease Activity Immune in a Confirmatory Cohort of SLE Patients [abstract]. Arthritis Rheumatol. 2020; 72 (suppl 10). https://acrabstracts.org/abstract/ability-of-innate-adaptive-and-tnf-superfamily-immune-pathways-to-characterize-disease-activity-and-inform-a-refined-lupus-disease-activity-immune-in-a-confirmatory-cohort-of-sle-patients/. Accessed .« Back to ACR Convergence 2020
ACR Meeting Abstracts - https://acrabstracts.org/abstract/ability-of-innate-adaptive-and-tnf-superfamily-immune-pathways-to-characterize-disease-activity-and-inform-a-refined-lupus-disease-activity-immune-in-a-confirmatory-cohort-of-sle-patients/