Session Information
Date: Tuesday, November 15, 2016
Title: ACR/ARHP Combined Abstract Session: Orthopedics and Rehabilitation
Session Type: ACR Concurrent Abstract Session
Session Time: 4:30PM-6:00PM
Background/Purpose: Obesity is a common risk factor for knee osteoarthritis (KOA). While it is intuitive that bariatric weight loss improves knee pain, it is not clear how much is due to decreased mechanical load vs metabolic changes.
Methods: Patients were screened for knee pain prior to sleeve gastrectomy, gastric bypass, or laparoscopic gastric banding. We required pain for ≥15 days/month and VAS pain ≥ 30, excluding lupus, inflammatory arthritis, crystal disease, psoriasis, and bilateral knee replacement. Enrolled patients took standing knee xrays for Kellgren-Lawrence (KL) grading. We measured BMI and used the Knee Injury and Osteoarthritis Outcome Score (KOOS) questionnaire at baseline and 1, 3, 6 and 12 months, calculating % excess weight loss (%EWL) and ΔKOOS. We collected blood at baseline and followup to study biomarkers for predicting KOOS scores.
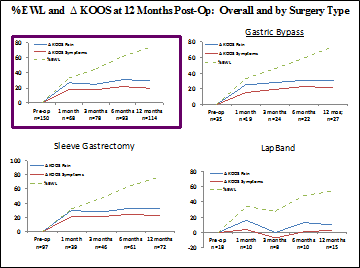
Results: Of 536 patients considering bariatric surgery, we found 308 with knee pain and enrolled 176 (91.5% female; BMI 43.6 kg/m2±7, 32-61; age 42 ±11, 18-73) well distributed in xray severity (KL0-4). For the 150 patients who had surgery, knee improvement paralleled weight loss at the followups. At 1 year, %EWL correlated well with ΔKOOS pain (R = .262, n = 114, p = 0.005), similar to other intervals and to other KOOS measures. The sleeve and bypass (n=72 and 27) vs banding (n=15) resulted in higher ΔKOOS pain at 1 year: 32.9 ±21.3 and 30.7 ±22.6 vs 10.2 ±21.4, p=0.001. Sleeve and bypass patients also achieved a higher % of their potential ΔKOOS pain improvement than did banding (65.2% and 60.1% vs 16.8% of remaining KOOS points to 100), and a higher % of patients improved to any degree (93.1% and 88.9% vs 66.7%). Radiographic severity did not predict ΔKOOS at 1 year, nor did the presence of key comorbidities. Patients lost weight in a near-linear fashion through 1 year (Fig. 1), but their KOOS improvements plateaued at 1 month. This held true in sleeve and bypass subgroups (with altered anatomy), while banding showed less consistent ΔKOOS despite a similar trend in %EWL. Baseline leptin levels in obese KOA were higher than non-obese KOA and non-obese/non-OA controls from other cohorts (100.2 ±61.9 vs 26.2 ±16.7 and 15.4±13.8, p<0.001). Similarly, IL-1Ra, a potential marker of OA progression, was much higher than non-obese KOA or controls (1123±940 vs 324.0±145.6 and 272±130.0, p<0.001). Within obese KOA, higher leptin levels predicted worse xrays (KL0/1 vs KL2/3/4, p = 0.037). After 1 year, mean leptin and IL1-Ra from obese KOA patients had decreased (p<0.001).
Conclusion: Bariatric surgery improves knee OA symptoms proportionally to %EWL. Most relief occurs during the 1st month before much weight loss, suggesting a metabolic impact beyond mechanical load reduction on joints – at least with the sleeve and bypass that alter digestive anatomy. Leptin and IL-1Ra serum levels are elevated in obese KOA vs non-obese KOA and controls – and fall after bariatric surgery which could contribute to knee pain relief.
To cite this abstract in AMA style:
Mukherjee T, Bomfim F, Wilder E, Browne L, Toth K, Aharon S, Lin J, La Rocca Vieira R, Ren-Fielding C, Parikh M, Abramson SB, Attur M, Samuels J. The Impact of Obesity on Knee Osteoarthritis Symptoms and Related Biomarker Profiles in a Bariatric Surgery Cohort [abstract]. Arthritis Rheumatol. 2016; 68 (suppl 10). https://acrabstracts.org/abstract/the-impact-of-obesity-on-knee-osteoarthritis-symptoms-and-related-biomarker-profiles-in-a-bariatric-surgery-cohort/. Accessed .« Back to 2016 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/the-impact-of-obesity-on-knee-osteoarthritis-symptoms-and-related-biomarker-profiles-in-a-bariatric-surgery-cohort/