Session Information
Session Type: ACR Abstract Session
Session Time: 2:30PM-4:00PM
Background/Purpose: Interferon (IFN) signaling is upregulated in dermatomyositis (DM) and thought to play a role in pathogenesis. An IFN gene signature in peripheral blood of DM patients correlates with skin disease and may predict disease activity. However, studies have not analyzed how IFN signaling differs across DM subtypes or how it is influenced by multiple organ system involvement. Autoantibodies in DM distinguish subsets of disease with unique clinical features and outcomes. We hypothesized that strength and clinical utility of the DM blood IFN signature depends on autoantibody subtype and other clinical factors. Utilizing a clinically phenotyped DM cohort, we sought to understand the utility of blood IFN gene signature as markers of skin disease severity across patients differing in autoantibody subtype, muscle disease, interstitial lung disease (ILD), and cancer status.
Methods: Stranded RNA sequencing was performed on 377 whole blood samples derived from 205 DM patients seen at the Stanford Outpatient Clinic. The least absolute shrinkage and selection operator (LASSO) method for linear regression selected optimal gene signatures and errors were calculated in an independent dataset. An IFN score was calculated by averaging the healthy-normalized FPKM values for a selection of genes. Skin disease activity was quantitated using the Cutaneous Dermatomyositis Activity and Severity Index activity score (CDASI-a). Linear regression and Pearson correlation analyzed the association between longitudinal CDASI-a and IFN scores, adjusted and stratified across antibody subtype, ILD, cancer, and muscle disease status.
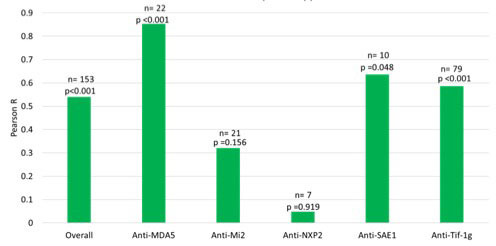
Results: We found that the blood IFN score is significantly elevated in the anti-MDA5 subtype compared to other subtypes (average 16.12, p < 0.001) [Figure 1]. Change in CDASI-a correlates most strongly with change in IFN score in the anti-MDA5 (R=0.85, p< 0.001) subtype, followed by anti-Tif-1g (R=0.59, p< 0.001) and anti-SAE1 (R=0.64, p =0.048) [Figure 2]. Conversely, the correlation is weak in anti-Mi2 and anti-NXP2 subtypes. These patterns persist after adjustment for ILD, muscle disease, cancer, and medications. The correlation is stronger when baseline IFN score is greater than 1.5 (R =0.59, p < 0.001). The correlation between IFN score and CDASI-a score is weaker in patients with active muscle disease and stronger in patients with active lung disease but is unaffected by cancer status.
Conclusion: Using a large prospective dataset of DM patients, we demonstrate that IFN-driven gene expression as an activity measure in DM is related to specific autoantibody subtypes. This relationship is also impacted by clinical factors such as ILD and muscle disease status. These results suggest that careful attention to antibody status and clinical factors could help inform interpretation of IFN biomarker data in future clinical trials in DM.
To cite this abstract in AMA style:
Tabata M, Sarin K, Page K, Huard C, Zhao S, Bennett D, Johnson J, Johnson K, Fiorentino D. Skin Disease Activity and Autoantibody Phenotype Are Major Determinants of Blood Interferon Signatures in Dermatomyositis [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/skin-disease-activity-and-autoantibody-phenotype-are-major-determinants-of-blood-interferon-signatures-in-dermatomyositis/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/skin-disease-activity-and-autoantibody-phenotype-are-major-determinants-of-blood-interferon-signatures-in-dermatomyositis/