Session Information
Session Type: Poster Session (Tuesday)
Session Time: 9:00AM-11:00AM
Background/Purpose: Hydroxychloroquine (HCQ) is the most well established therapy for SLE, as it provides several beneficial properties, such as favorable effects on lipid profile, reduced cardiovascular (CV) risk and reduction in flares. However, one of the main concerns about HCQ use is retinopathy. In this regard, the American Academy of Ophthalmology (AAO) recently recommended the maximum daily dose of 5.0 mg/kg of real weight but this recommendation is only focused on retinal safety and there are no data evaluating if reduced doses would sustain the beneficial effects of this drug with regard to lipid profile and flare rates. Therefore, the aim of this study was to prospectively assess lipid profile and flare rate in high-risk retinopathy stable LN patients using low dose HCQ AAO recommendation.
Methods: Eighty-two stable LN patients (6-months before entry), ≥5 years of HCQ were evaluated at entry and after 6-months (6M). None of them had retinopathy (spectral-domain optical coherence tomography). HCQ blood levels were determined by liquid chromatography-mass spectrometry. Fasting lipoproteins/blood glucose levels were determined et entry and 6M. Flare was defined as increase in SLEDAI-2K≥3 and/or change in therapy. We also evaluated retrospectively lipid profile, high CV risk and flare rates before HCQ dose reduction according to AAO and compared to the same patients at the end of prospective analysis.
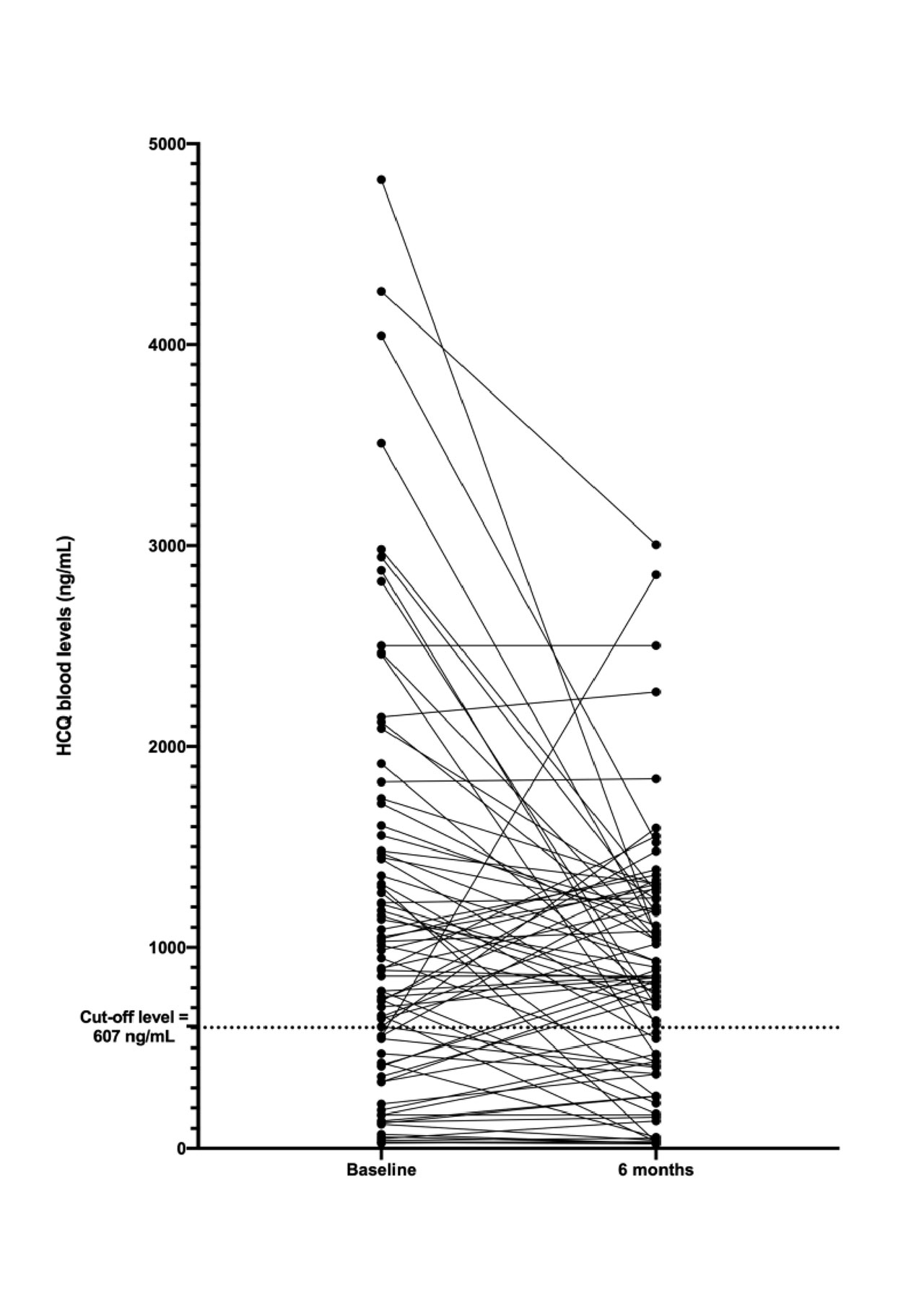
Results: Total/LDL/HDL cholesterol and triglyceride levels remained stable throughout the study (p >0.05) and no significant differences were observed in frequencies of high CV risk levels (p >0.05). Nine of 82 LN patients (10.97%) had flare during the 6-months follow-up. HCQ blood levels were significantly lower in patients with compared to those without flare [220.4(32.1-1471.7) vs. 986.6(26.1-4265.0)ng/mL, p=0.015]. Comparisons of other baseline characteristics demonstrated similar demographic data, disease parameters and concomitant treatment of LN patients with and without flare. An HCQ blood level cut-off of 607ng/ml was a predictor of 6-months flare [OR=5.67, 95%CI 1.34-24.03; sensitivity 78%, specificity 71%, p=0.002). Of note, retrospective analysis revealed comparable lipoprotein and glycemia levels as well as, frequencies of high CV risk and flare rates (9% vs. 10%, p=0.751) before and after AAO dose reduction (Table 1). HCQ blood levels were classified in low (< 607 ng/mL) or adequate (≥607 ng/mL) according to the cut-off level. At 6M evaluation, blood HCQ levels remained within the same range in 67 patients (82%), whereas it turned from low to adequate in six and from adequate to low in nine patients (McNemar’s test, p= 0.606) (Figure 1).
Conclusion: We provide novel evidence that AAO HCQ low dose recommendation in stable LN patients with high-risk retinopathy is not associated with a deleterious effect on lipid profile or increased flare rates. This finding supports the notion that this new regimen retains the beneficial effect of HCQ with the advantage of a lower long-term cumulative dose and toxicity.
To cite this abstract in AMA style:
Pedrosa T, Pasoto S, Yuki E, Aikawa N, Borba E, Ferreira Filho J, Carricondo P, Zanetti C, Conde P, Fontoura N, Romano P, Carvalho V, Silva C, Bonfa E. Prospective Evaluation of American Academy of Ophthalmology Low Dose Hydroxychloroquine Recommendation in Stable Lupus Nephritis with High-Risk Retinopathy: Lipid Profile and Flare Rates [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/prospective-evaluation-of-american-academy-of-ophthalmology-low-dose-hydroxychloroquine-recommendation-in-stable-lupus-nephritis-with-high-risk-retinopathy-lipid-profile-and-flare-rates/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/prospective-evaluation-of-american-academy-of-ophthalmology-low-dose-hydroxychloroquine-recommendation-in-stable-lupus-nephritis-with-high-risk-retinopathy-lipid-profile-and-flare-rates/