Session Information
Session Type: ACR Abstract Session
Session Time: 4:30PM-6:00PM
Background/Purpose: Secukinumab (SEC) is an interleukin-17 inhibitor used to treat patients with axial spondylarthritis (axSpA) and psoriatic arthritis (PsA). Drug survival is often used as a proxy for treatment effectiveness and safety. We aim to assess SEC survival in routine clinical practice and to identify survival predictors associated.
Methods: We conducted a retrospective, longitudinal, observational, multicentric study including all patients with axSpA or PsA who received at least 1 injection of SEC between July 2016 and April 2019. We collected demographic and clinical characteristics, onset date, initial dosage and dosage modification of SEC, previous biologic Disease-modifying antirheumatic drugs (bDMARDs) and concomitant treatments. We classified the reason of discontinuation in lack of effectiveness, adverse events and others. Potential predictors evaluated were baseline age, gender, smoking status, previous bDMARDs and concomitant treatment. Among the axSpA cohort, evidence of radiographic sacroiliitis, MRI sacroiliitis or elevated CRP were also assessed as potential predictors. Drug survival was analyzed by the Kaplan-Meier method and differences of survival between groups was assessed by log-rank test.
Results: The main characteristics of the 556 patients (pts) included were the following: 338 (61%) axSpA , 183 (33%) PsA, 324 (58%) female, mean age 47 +/- 12 years, 221 (40%) smokers,160 (29%) radiographic sacroiliitis, 238 (43%) MRI sacroiliitis, 206 (37%) elevated CRP, 239 (43%) HLA B27 positive, mean BASDAI 48,9 +/-27%. SEC was associated to methotrexate (MTX) in 140 pts (25%) and was the first line bDMARD in 56 patients (10%).
The median drug survival (MDS) of SEC was 76 weeks (w) (interquartile range [65-88]). At 52w, 245 pts (60%) SpA were still treated with SEC. Among reasons of discontinuation, 172 (68%) pts discontinued SEC for lack of effectiveness, 46 (18%) for adverse events and 13 (5.1%) for others.
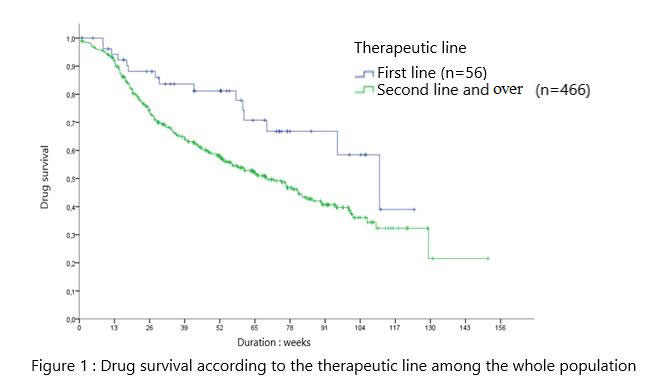
First line SEC administration was associated with a longer survival versus second line and more: 111w [83-138] vs 69w [57-81] (p=0.01) (figure 1). MDS was not significantly different according to gender, disease, MTX combo, elevated CRP, axSpA vs PsA and smoking status.
Among the axSpA pts, absence of radiographic or MRI sacroiliitis and normal CRP did not modify significantly SEC survival (p=0.85) (figure2).
Conclusion: In routine clinical practice, SEC median survival was 76 weeks. Fist line administration was associated with improved SEC retention. Lack of effectiveness was the most common reason of discontinuation.
To cite this abstract in AMA style:
Flachaire B, Letarouilly J, Labadie C, Cohen N, Pradel V, Sellam J, Richette P, Dieudé P, Claudepierre P, Fautrel B, Houvenagel E, Nguyen C, Guyot M, Segaud N, Maury F, Marguerie L, Deprez X, Salmon J, Baudens G, Gervais E, Miceli-Richard C, Chary-Valckenaere I, Lafforgue P, Loeuille D, Richez C, Flipo R, Pham T. Predictors of Survival of Secukinumab Treatment in a Multicenter Cohort of 556 Spondylarthritis [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/predictors-of-survival-of-secukinumab-treatment-in-a-multicenter-cohort-of-556-spondylarthritis/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/predictors-of-survival-of-secukinumab-treatment-in-a-multicenter-cohort-of-556-spondylarthritis/