Session Information
Session Type: Poster Session D
Session Time: 1:00PM-3:00PM
Background/Purpose: Lyme arthritis (LA), caused by infection with Borrelia burgdorferi (Bb), is characterized by proliferative synovitis accompanied by dysregulated autoimmune Th1 responses. Fibroblast-like synoviocytes (FLS) are the most abundant cell in the synovial lesion and express markers of immune activation, including MHC class II molecules. Bb cell wall peptidoglycan (PG) is a persistent pathogen-associated molecular pattern (PAMP) in joints of LA patients and persists months to several years after antibiotic therapy and apparent spirochetal killing, called post-infectious LA. We hypothesize that PG acts as an immune adjuvant to activate MHCII+ FLS and enhance Th1 activation in synovial tissue during infection and in post-infectious LA.
Methods: FLS were isolated and purified from tibiotarsal ankles of C57BL/6 mice and stimulated with IFN-gamma, PG, or both. Wild-type macrophages and macrophages and FLS from mice deficient in the PG-sensing pattern recognition receptor NOD2 were used as controls. Immune responses were measured by flow cytometry and cytokine analyses. T cell activation was analyzed by coculturing MHCII+ FLS with CFSE-labeled naive OT-II T cells supplemented with the cognate OVA323-339 peptide or irrelevant OVA257-264 peptide as control. T cell activation was measured using a proliferation assay and T cell cytokine analysis.
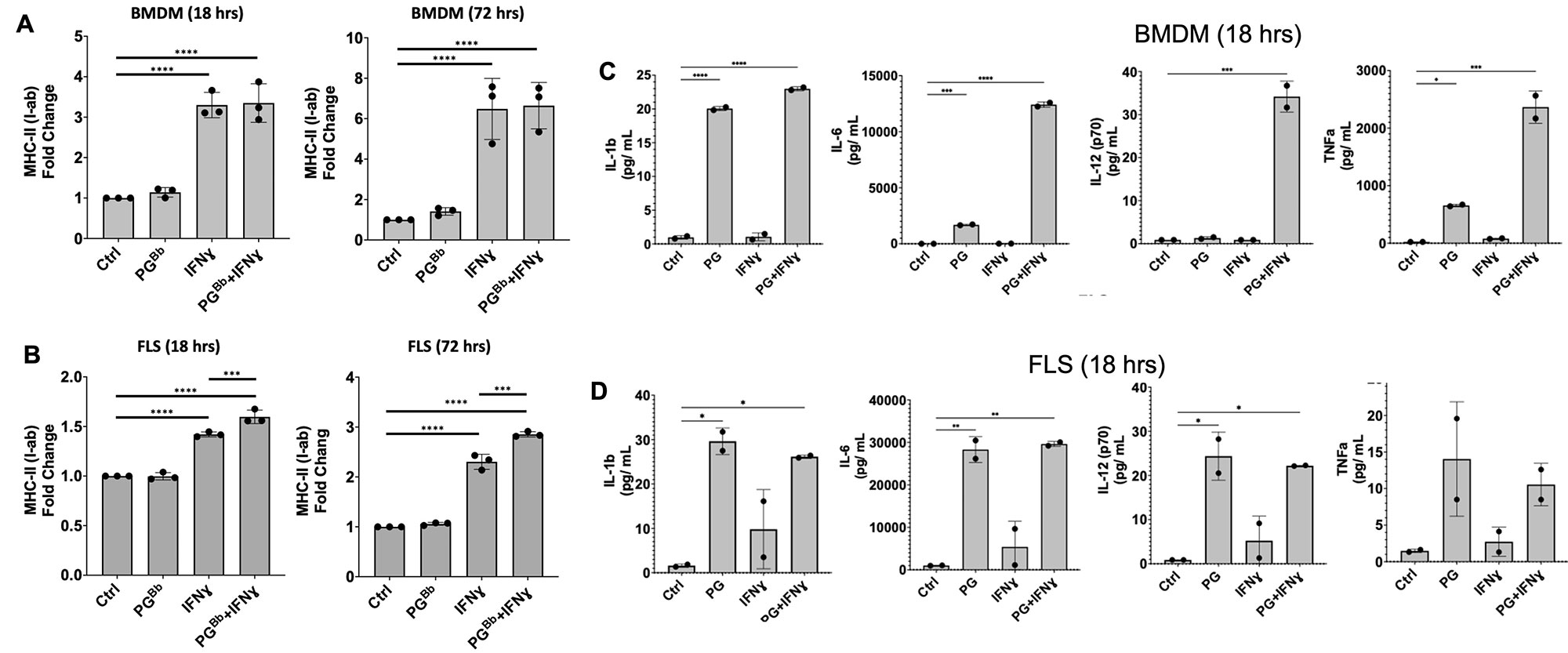
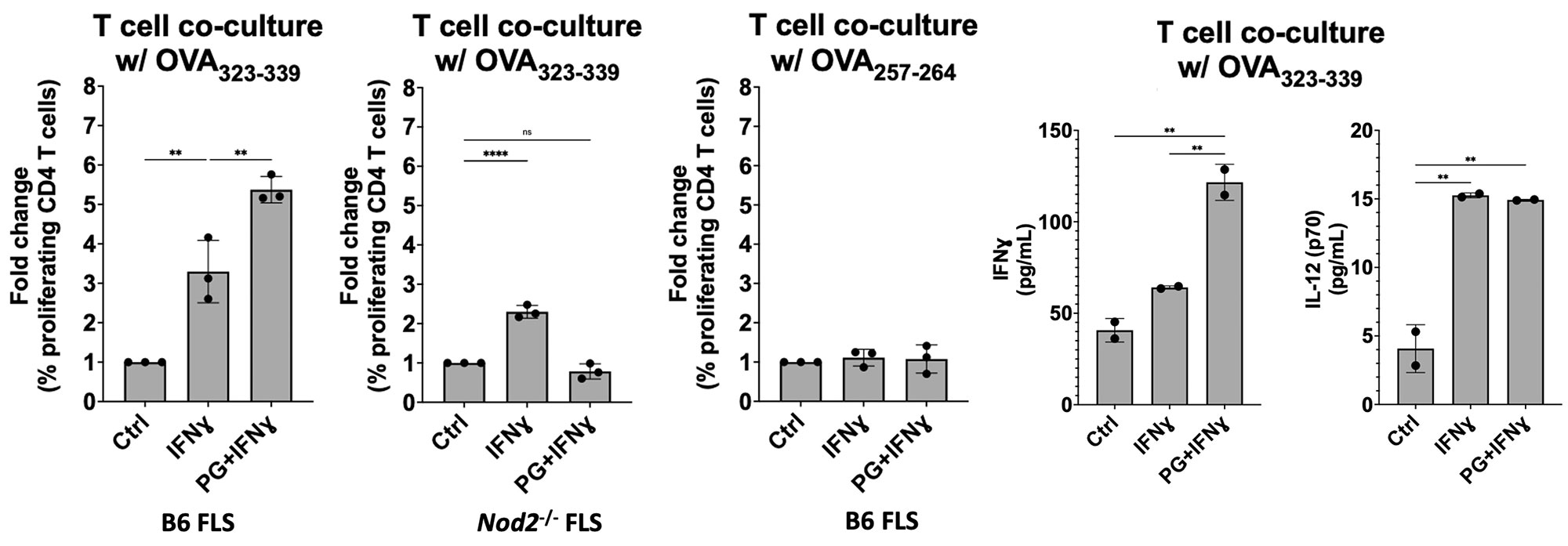
Results: FLS stimulated with IFN-gamma expressed MHCII and pro-inflammatory cytokines IL-1, IL-6, and IL-12, consistent with our previous results using primary human FLS. FLS stimulated with both PG and IFN-gamma expressed significantly higher levels of MHCII and inflammatory cytokines, compared with IFN-gamma stimulation alone (Figure 1). Naive OT-II T cells, which only recognize the MHCII peptide OVA323-339, proliferated 3-fold compared with media alone control when co-cultured with FLS primed with IFN-gamma and supplemented with the OVA323-339 peptide. FLS supplemented with the irrelevant OVA257-264 peptide did not elicit OT-II T cell proliferation. FLS primed with both IFN-gamma and PG induced a 5-fold increase in OT-II proliferation, compared with media alone control. T cell supernatants contained high levels of IFN-gamma, indicating Th1 polarization of activated CD4+ T cells. FLS lacking NOD2 failed to induce CD4+ T cell proliferation (Figure 2).
Conclusion: These data demonstrate that PG acts as an immune adjuvant by enhancing the ability of MHCII+ FLS to induce CD4+ T cell proliferation in a peptide-dependent and NOD2-dependent manner. This response recapitulates the autoimmune Th1 responses observed in synovial tissue of post-infectious LA patients. This study helps us understand the critical role MHCII+ FLS and bacterial cell wall debris play in driving persistent inflammatory T cell responses within the synovial lesion, even in the absence of active infection. These results also shed light on development of autoimmunity in other inflammatory joint diseases where bacteria are thought to be an autoimmune trigger.
To cite this abstract in AMA style:
Rouse J, Wahhab A, Danner R, Jutras B, Edelstein A, Strle K, Lochhead R. Peptidoglycan Amplifies CD4+ T Cell Activation by MHCII+ Fibroblast-like Synoviocytes in an in Vitro Model of Lyme Arthritis [abstract]. Arthritis Rheumatol. 2022; 74 (suppl 9). https://acrabstracts.org/abstract/peptidoglycan-amplifies-cd4-t-cell-activation-by-mhcii-fibroblast-like-synoviocytes-in-an-in-vitro-model-of-lyme-arthritis/. Accessed .« Back to ACR Convergence 2022
ACR Meeting Abstracts - https://acrabstracts.org/abstract/peptidoglycan-amplifies-cd4-t-cell-activation-by-mhcii-fibroblast-like-synoviocytes-in-an-in-vitro-model-of-lyme-arthritis/