Session Information
Session Type: Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose: Cytokine Release Syndrome (CRS) or Macrophage Activation Syndrome (MAS) is a life threating hyperinflammatory condition that can complicate rheumatic disease as well as infections like SARS-CoV-2 (SARS-CoV-2-CRS). Immunomodulators are traditionally used to treat MAS in SLE, Kawasaki disease, and systemic JIA, but have the potential to treat SARS-CoV-2-CRS. An institution-mandated rheumatology SARS-CoV-2-CRS management algorithm was developed for 2 health systems with use of tocilizumab (TOC) and anakinra (ANA) as first and second-line agents. The objective of this study is to determine characteristics of patients from a SARS-CoV-2-CRS cohort with the overall goal to understand the clinical disease course of these patients and the impact of biologic management.
Methods: A retrospective chart review was completed on SARS-CoV-2 patients at an academic medical center and a county hospital for whom rheumatology consultations were completed for CRS evaluation from March 15-May 30, 2020. Both institutions shared the same electronic medical record system. Patients meeting CRS diagnostic criteria who were treated with biologic therapy were compared to those without CRS not given biologic therapy for the following outcomes: survival, deaths, infections, intensive care unit (ICU) admissions and duration, length of hospitalization, thrombosis and trends in laboratory markers including blood cell counts, liver enzymes, and acute phase reactants.
Results: Out of 150 patients with SARS-CoV-2 referred for rheumatology consultation, 71 (47%) were diagnosed with CRS and were treated with the following biologics: TOC only in 63 (89%), ANA only in 1 (1%), abatacept only in 1 (1%), and 6 (8%) patients who received both TOC and ANA separately. Steroids were given in 64 (90%) CRS patients. Comparing baseline characteristics of all 150 patients (Table 1), CRS patients required more ICU admissions (61%), but had similar need for ventilator support (23%). Biologic treatment resulted in improved ferritin, platelets, and ESR; yet, other laboratory tests remained similar or worse 3-5 days after the first dose (Table 2). Death occurred in 20 (28%) while 36 (51%) were discharged from the hospital (Table 3). Bacterial co-infection at time of diagnosis was noted in 10 (14%); 10 (14%) patients developed a second co-infection after biologic treatment.
Conclusion: SARS-CoV-2-CRS patients had similar baseline comorbidities to patients without CRS except for co-infections at time of diagnosis that were more common in the latter group. Within the time frame laboratory tests were reviewed, only the ferritin, platelets, and ESR improved after biologic treatment, but overall, patients with CRS had more critical laboratory test results compared to those without CRS. Death was noted to be two-fold higher in CRS patients. All deceased were in the ICU at the time, suggesting a sicker population at greater risk for death. Additional studies are needed including a longitudinal evaluation of trends in laboratory tests post-biologic use in consideration of drug pharmacokinetics and more robust cytokine profiling. Furthermore, outcomes in CRS patients that received biologics should be compared to a control arm that did not receive biologics.
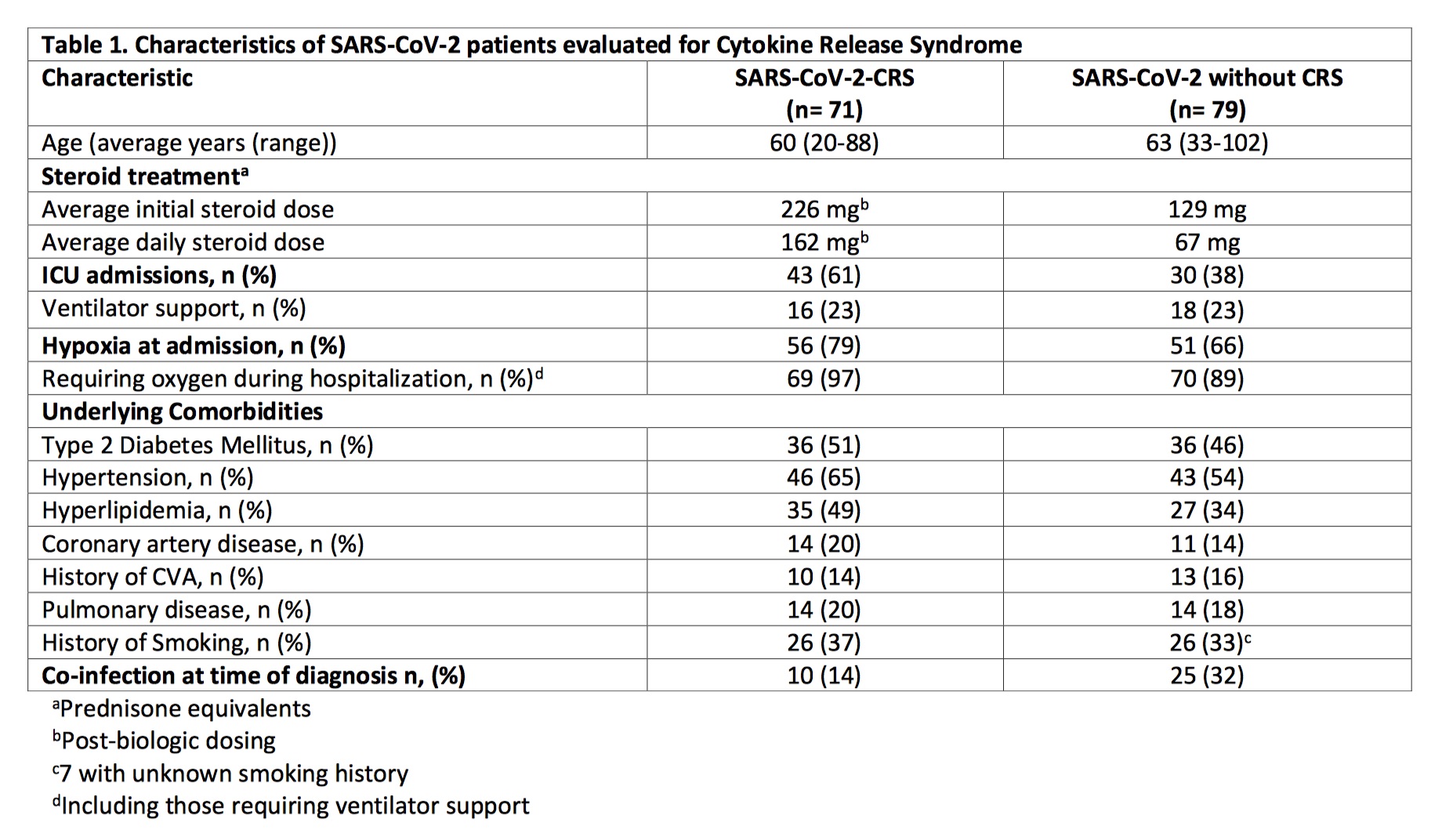 Characteristics of SARS-CoV-2 patients evaluated for Cytokine Release Syndrome
Characteristics of SARS-CoV-2 patients evaluated for Cytokine Release Syndrome
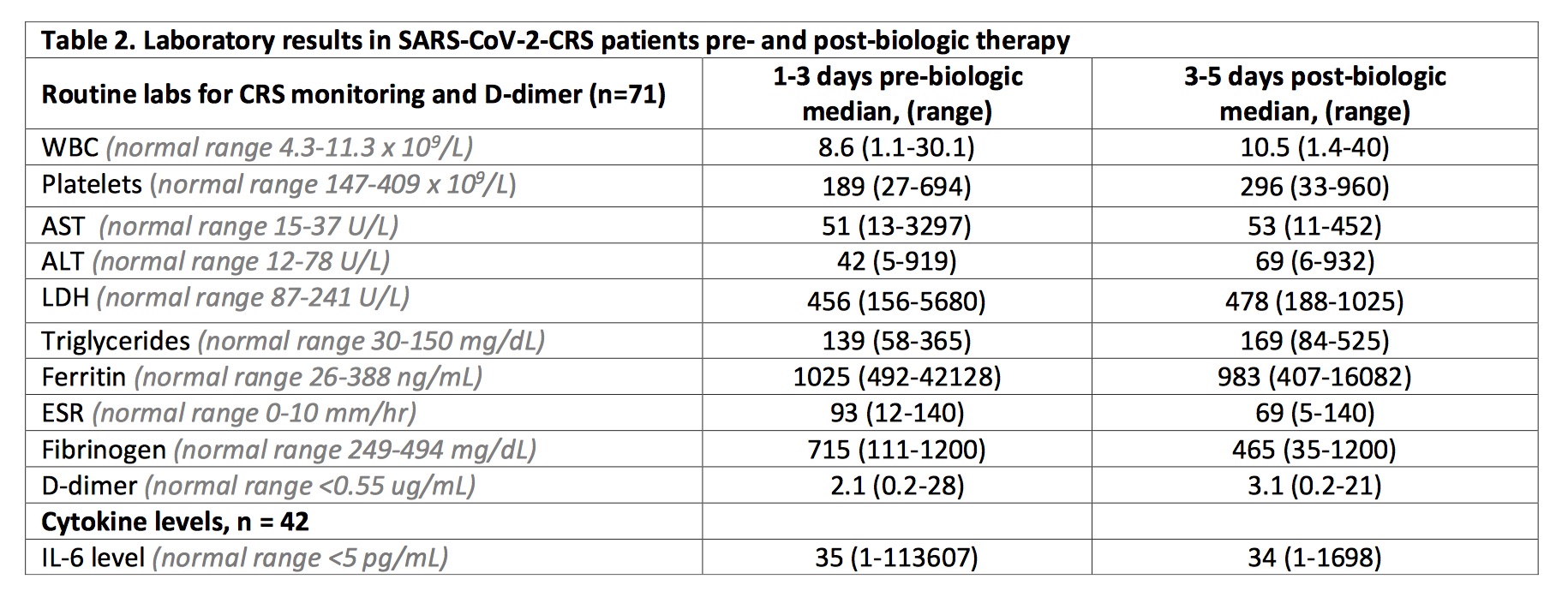 Laboratory results in patients with Cytokine Release Syndrome pre- and post-biologic therapy
Laboratory results in patients with Cytokine Release Syndrome pre- and post-biologic therapy
 Outcomes in SARS-CoV-2-CRS treated with biologic and Non-SARS-CoV-2-CRS not treated with biologic
Outcomes in SARS-CoV-2-CRS treated with biologic and Non-SARS-CoV-2-CRS not treated with biologic
To cite this abstract in AMA style:
Chiruvolu N, Karim M, Injean P, Lee S, Torralba K, Downey C, Hojjati M, Doo L, Jose D, Panikkath D, Yu M, Lafian A, Cabling M, Sandhu V. Cytokine Storm: Outcomes in SARS-CoV-2 Patients Treated with Biologics in a Rheumatology Cohort [abstract]. Arthritis Rheumatol. 2020; 72 (suppl 10). https://acrabstracts.org/abstract/cytokine-storm-outcomes-in-sars-cov-2-patients-treated-with-biologics-in-a-rheumatology-cohort/. Accessed .« Back to ACR Convergence 2020
ACR Meeting Abstracts - https://acrabstracts.org/abstract/cytokine-storm-outcomes-in-sars-cov-2-patients-treated-with-biologics-in-a-rheumatology-cohort/
