Session Information
Session Type: Abstract Session
Session Time: 4:15PM-4:30PM
Background/Purpose: The identification of intrarenal pathological processes is key to develop better diagnostic and treatment strategies in lupus nephritis (LN). But the direct comprehensive study of renal tissue can be limited by tissue degradation, availability, and cell survival. We employed urine proteomics to define the molecular pathways involved in proliferative LN
Methods: We quantified 1200 biomarkers (Kiloplex, RayBiotech) in urine samples collected on the day of (73%) or within 3 weeks (27%) of kidney biopsy in SLE patients with urine protein to creatinine ratio on random or 24 hr collection of > .5. Urine proteomic profiles were analyzed with respect to lupus nephritis histological features.
Results: A total of 195 patients were included: 138 (71%) had a proliferative histological class (III or IV +/- V), 57 (29%) pure membranous (V). There were 21 (FDR 1%) differentially abundant urinary proteins in proliferative compared to pure membranous LN (Figure 1A). These included several neutrophil granule proteins (Figure 1B) in addition to previously reported biomarkers such as IL-16 and CD163. Unsupervised clustering based on the proliferative LN signature identified 3 groups characterized by low, medium or high protein abundance (Figure 2). Higher proliferative signature abundance (right cluster) was associated with higher histological activity (NIH Activity Index). Immunofluorescence revealed an abundant MPO+ neutrophil infiltrate in proliferative LN (Figure 3).
Conclusion: Proliferative LN was associated with a urinary neutrophil degranulation signature, especially in patients with higher histological activity. Neutrophil activity could be non-invasively monitored to assist with the diagnosis of proliferative LN. These findings implicate neutrophils in LN activity and pathogenesis, nominate urinary neutrophil signatures as noninvasive biomarkers, and support the study of treatment targeted to neutrophils.
 Figure 1. Urine proteomic profile of proliferative LN. (A) Volcano plot displaying the log fold change (FC) and adjusted p values of the differential abundance of 1200 urinary proteins. (B) Pathway enrichment analysis (Gene Ontology and Reactome) of the proteins enriched (FDR < 1%) in proliferative LN. Odds ratios based on the hypergeometric test are displayed.
Figure 1. Urine proteomic profile of proliferative LN. (A) Volcano plot displaying the log fold change (FC) and adjusted p values of the differential abundance of 1200 urinary proteins. (B) Pathway enrichment analysis (Gene Ontology and Reactome) of the proteins enriched (FDR < 1%) in proliferative LN. Odds ratios based on the hypergeometric test are displayed.
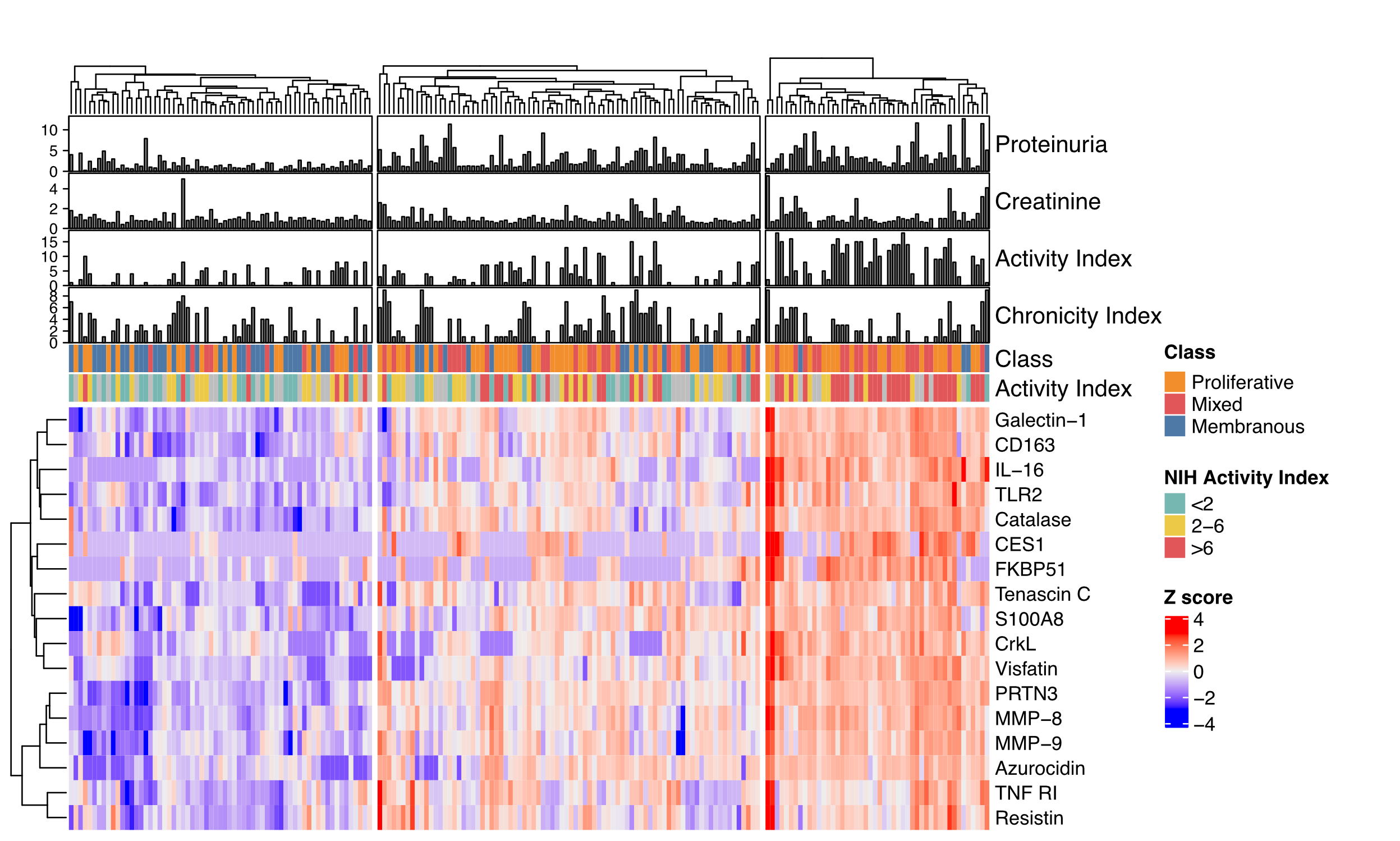 Figure 2. Higher urinary neutrophil signature is associated with higher lupus nephritis activity. Heatmap of the urinary protein differentially abundant in proliferative LN. Hierarchical clustering based on protein abundance identified 3 groups. Proteinuria in mg protein/mg creatinine.
Figure 2. Higher urinary neutrophil signature is associated with higher lupus nephritis activity. Heatmap of the urinary protein differentially abundant in proliferative LN. Hierarchical clustering based on protein abundance identified 3 groups. Proteinuria in mg protein/mg creatinine.
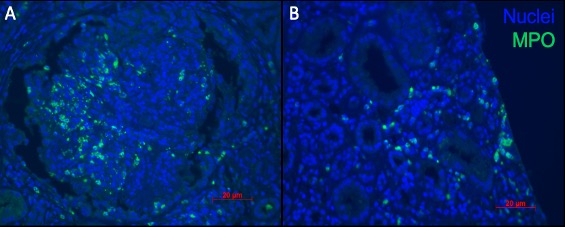 Figure 3. Neutrophil infiltrate in proliferative lupus nephritis. Immunofluorescence imaging of glomerular (A) and tubulointerstitial (B) neutrophil infiltration in a patient with class IV lupus nephritis. Myeloperoxydase (MPO) in green and DAPI in blue.
Figure 3. Neutrophil infiltrate in proliferative lupus nephritis. Immunofluorescence imaging of glomerular (A) and tubulointerstitial (B) neutrophil infiltration in a patient with class IV lupus nephritis. Myeloperoxydase (MPO) in green and DAPI in blue.
To cite this abstract in AMA style:
Fava A, Li J, Goldman D, Antiochos B, Monroy-Trujillo J, Fine D, Atta M, Buyon J, Guthridge J, James J, Petri M, (AMP) RA/SLE Network A. A Neutrophil Degranulation Signature Identifies Proliferative Lupus Nephritis [abstract]. Arthritis Rheumatol. 2021; 73 (suppl 9). https://acrabstracts.org/abstract/a-neutrophil-degranulation-signature-identifies-proliferative-lupus-nephritis/. Accessed .« Back to ACR Convergence 2021
ACR Meeting Abstracts - https://acrabstracts.org/abstract/a-neutrophil-degranulation-signature-identifies-proliferative-lupus-nephritis/
