Session Information
Session Type: Late-Breaking Abstract Session
Session Time: 4:00PM-6:00PM
Background/Purpose: Tofacitinib is an oral JAK inhibitor that is being investigated for JIA. Here we assess the efficacy and safety of tofacitinib in patients (pts) with JIA.
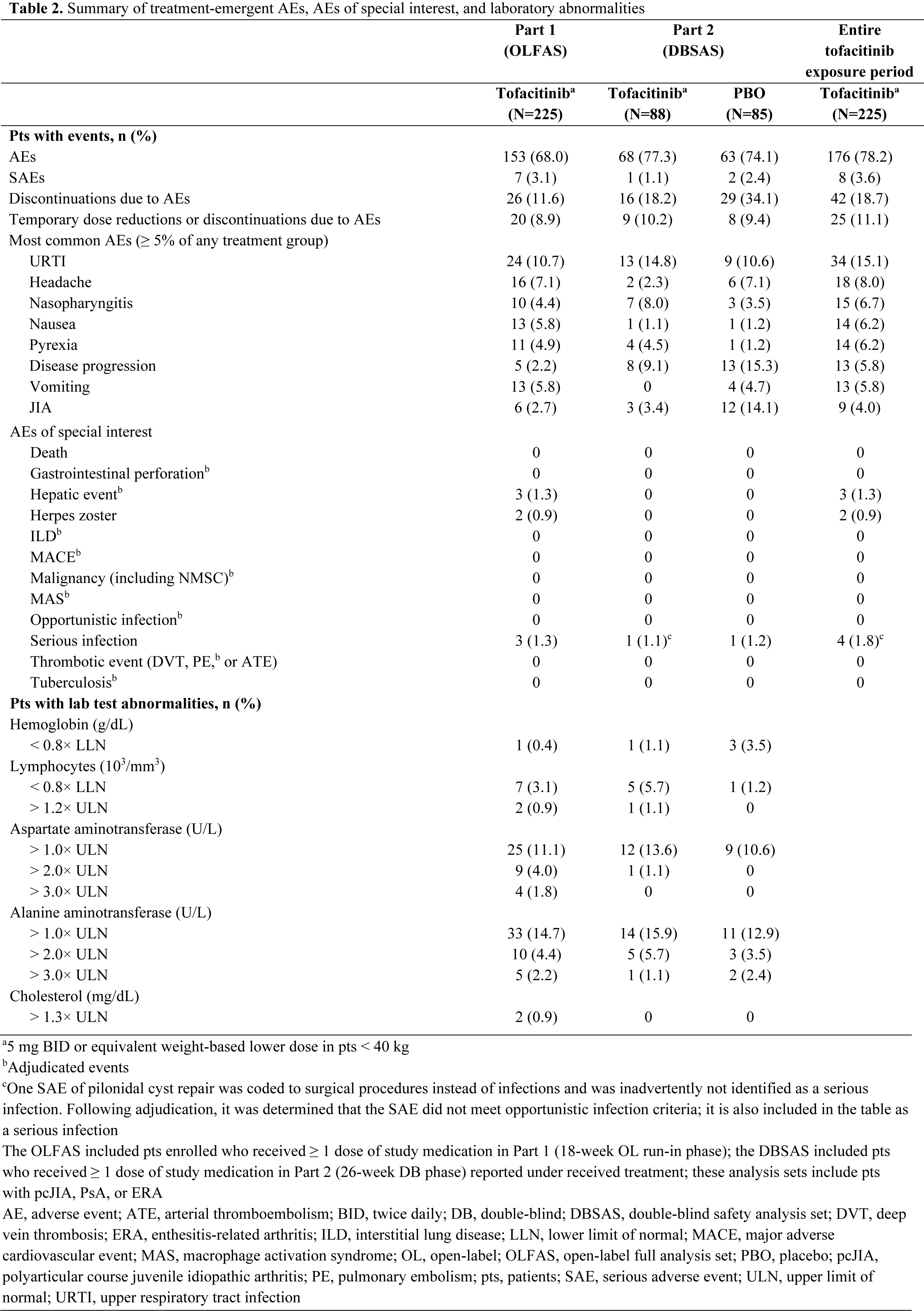
Methods: This was a Phase 3, randomized, double-blind (DB), placebo (PBO)-controlled withdrawal study in pts aged 2 to < 18 years with polyarticular course JIA (pcJIA), PsA or enthesitis-related arthritis (ERA) (NCT02592434). In the 18-week, open-label (OL), run‑in phase (Part 1), pts received tofacitinib. Pts achieving ≥ JIA ACR30 response1 at Week (W)18 were randomized 1:1 in the DB phase (W18−44; Part 2) to continue receiving tofacitinib or withdraw tofacitinib and newly receive PBO. Tofacitinib was administered according to body weight: 2−4 mg BID oral solution in pts < 40 kg; 5 mg BID tablet or oral solution in pts ≥ 40 kg. The primary endpoint was occurrence of disease flare2 by W44 (W26 of Part 2). Key secondary endpoints were JIA ACR50/30/70 response1 rates and change (∆) from Part 2 baseline (BL) in Childhood Health Assessment Questionnaire Disability Index (CHAQ-DI) at W44. The primary and key secondary endpoints were type 1 error-controlled with statistical significance declared at p < 0.05. Other efficacy endpoints included time to disease flare in Part 2, ∆ from Part 1 BL in JIA ACR core set variables1 at W44, and ∆ from Part 2 BL in Juvenile Arthritis Disease Activity Score (JADAS27-CRP) during Part 2. Pts with PsA or ERA were excluded from these efficacy analyses and analyzed separately. Time to flare was analyzed by the Kaplan-Meier method. Safety was evaluated throughout the study.
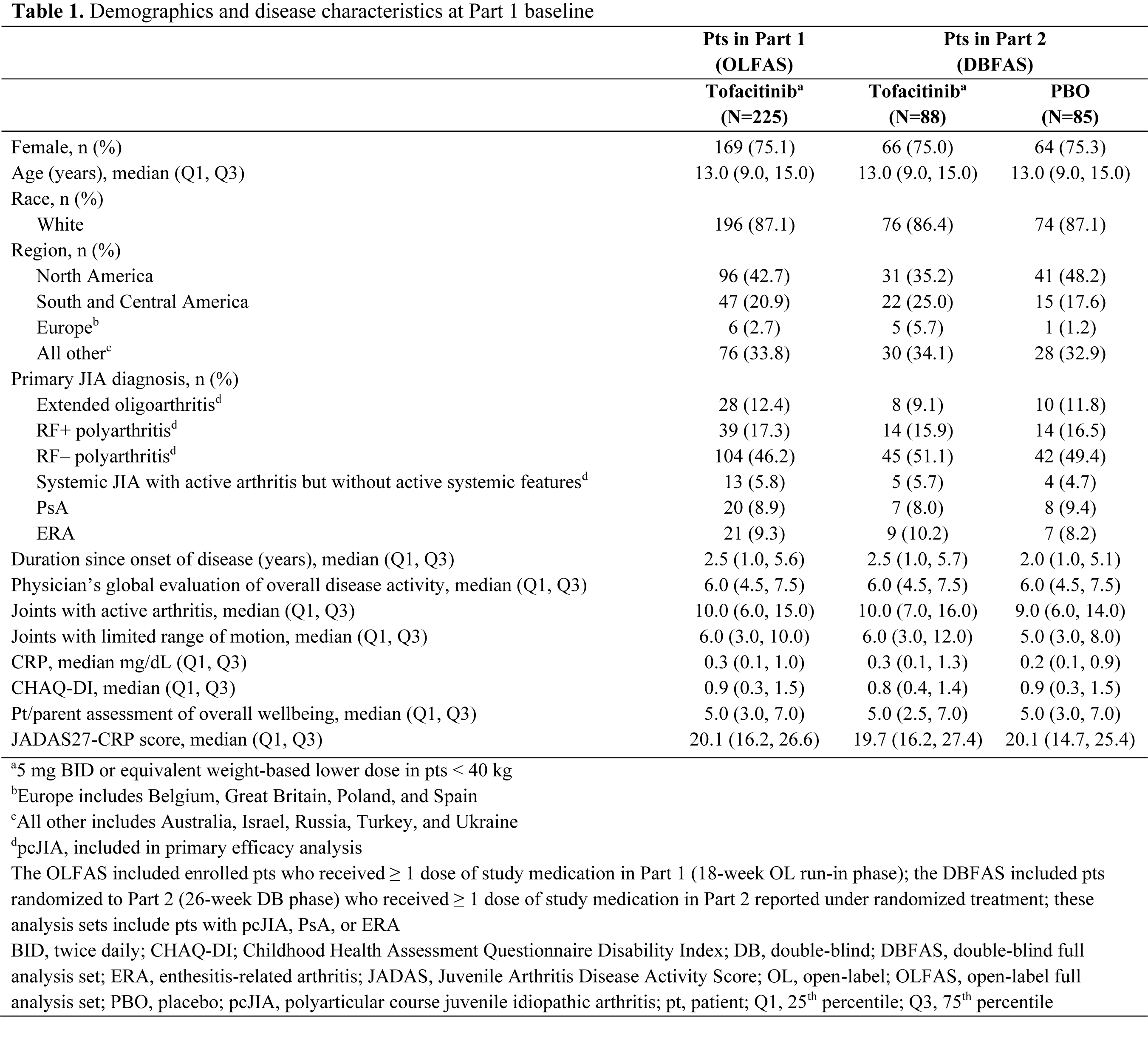
Results: 225 pts with pcJIA (n=184), PsA (n=20) or ERA (n=21) were enrolled and received OL tofacitinib in Part 1 (Table 1). At W18, 173/225 (76.9%) pts entered Part 2 (pcJIA [n=142], PsA [n=15], ERA [n=16]). In pts with pcJIA, occurrence of disease flare in Part 2 was significantly lower with tofacitinib (29.2%) vs PBO (52.9%) by W44 (p=0.0041; primary endpoint; Figure 1a). JIA ACR50/30/70 response rates (Figure 1b) and improvement from Part 2 BL in CHAQ-DI (Figure 1c) at W44 were greater with tofacitinib vs PBO. Time to disease flare was greater with tofacitinib vs PBO in Part 2 (Figure 1d). Tofacitinib had a greater effect vs PBO in reducing signs and symptoms of pcJIA, in terms of ∆ from Part 1 BL in JIA ACR core set variables at W44 (Figure 1e). From early time points in Part 2, disease activity (assessed by JADAS27-CRP) worsened with PBO but remained stable with tofacitinib (Figure 1f). Safety was generally similar in pts receiving tofacitinib or PBO (Table 2): 77.3% and 74.1% had adverse events (AEs); 1.1% and 2.4% had serious AEs. There were no cases of death, opportunistic infection or tuberculosis.
Conclusion: In pts with pcJIA, treatment with tofacitinib vs PBO resulted in significantly fewer disease flares, improved time to flare, improvements in disease signs and symptoms and physical functioning, and a sustained clinically meaningful improvement in disease activity. The safety profile of tofacitinib was consistent with that in adults with RA.
- Giannini EH et al. Arthritis Rheum 1997; 40: 1202-1209.
- Brunner HI et al. J Rheumatol 2002; 29: 1058-1064.
Acknowledgments: Study sponsored by Pfizer Inc. Medical writing support was provided by Sarah Piggott of CMC Connect and funded by Pfizer Inc.
To cite this abstract in AMA style:
Brunner H, Synoverska O, Ting T, Abud Mendoza C, Spindler A, Vyzhga Y, Marzan K, Keltsev V, Tirosh I, Imundo L, Jerath R, Kingsbury D, Sozeri B, Vora S, Prahalad S, Zholobova E, Butbul Aviel Y, Chasnyk V, Lerman M, Nanda K, Schmeling H, Tory H, Uziel Y, V. Tofacitinib for the Treatment of Polyarticular Course Juvenile Idiopathic Arthritis: Results of a Phase 3 Randomized, Double-blind, Placebo-controlled Withdrawal Study [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/tofacitinib-for-the-treatment-of-polyarticular-course-juvenile-idiopathic-arthritis-results-of-a-phase-3-randomized-double-blind-placebo-controlled-withdrawal-study/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/tofacitinib-for-the-treatment-of-polyarticular-course-juvenile-idiopathic-arthritis-results-of-a-phase-3-randomized-double-blind-placebo-controlled-withdrawal-study/

.png)