Session Information
The 2020 Pediatric Rheumatology Symposium, originally scheduled for April 29 – May 2, was postponed due to COVID-19; therefore, abstracts were not presented as scheduled.
Date: Friday, May 1, 2020
Title: Poster Session 2
Session Type: ACR Abstract Session
Session Time: 5:00PM-6:00PM
Background/Purpose: The introduction of biologic medications has revolutionized the care of children with systemic juvenile idiopathic arthritis (SJIA). Differences in treatment approaches among different countries and how they have changed in the modern era of biologic medications have not been well studied. We sought to contrast the presenting features, initial treatment and 12-month outcomes of SJIA patients in pediatric rheumatology Registries in the United States, Canada, United Kingdom, Portugal, Sweden, Denmark, Turkey, Germany, Norway and Finland using individual country registry data.
Methods: Data were extracted locally from the 10 JIA Registries. Disease manifestations collected within nine weeks of first visit with a pediatric rheumatologist (PR) or at diagnosis were included as presenting features. Data on medication use within first year of diagnosis were collected. Clinical outcomes, including the Physician Global Assessment (PGA), active joint count (AJC) and current glucocorticoid (GC) use, were collected at the 12±3 month visit following diagnosis. To assess modern era treatment approaches, we compared data for children treated before and after 2012. Weighted means were used to adjust for varying numbers of patients per Registry.
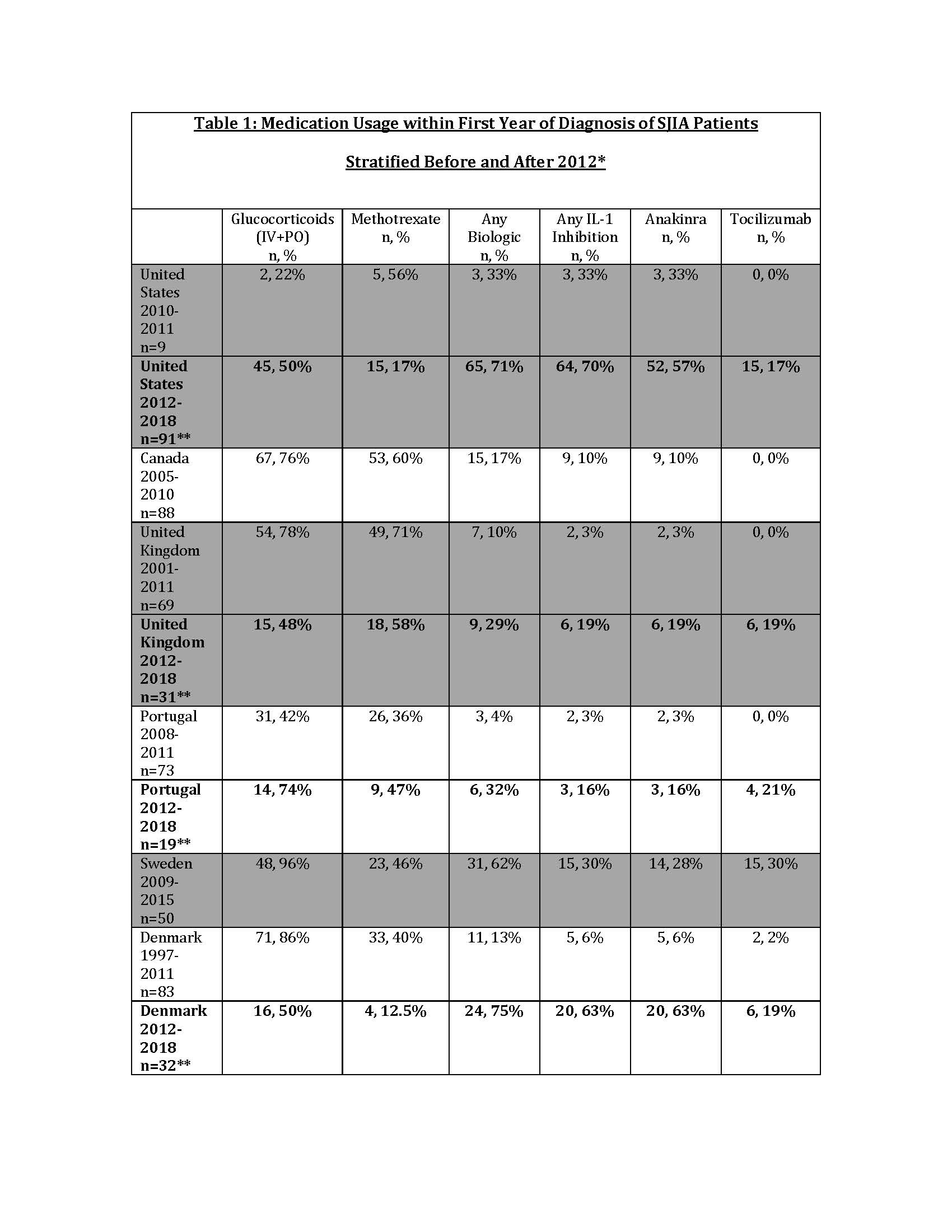
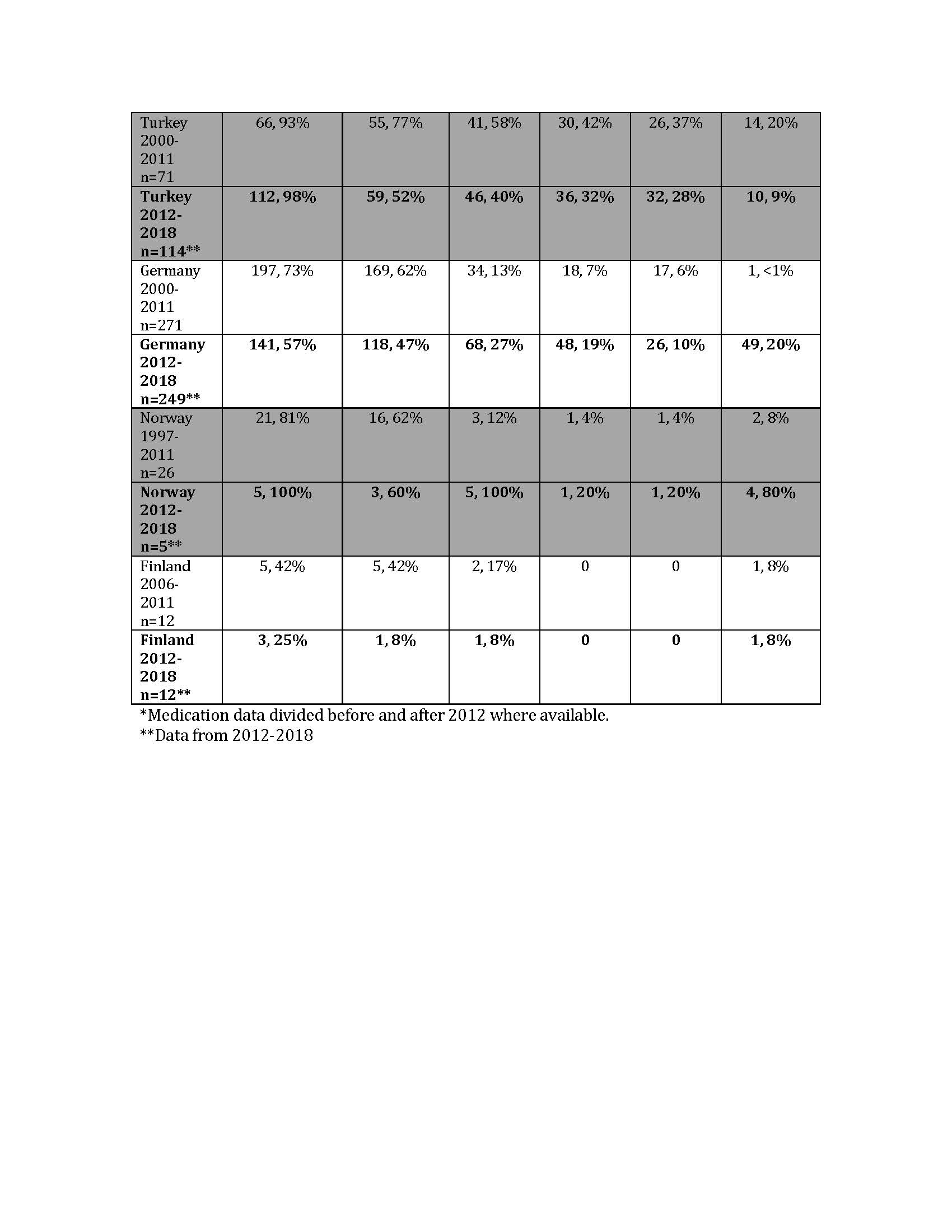
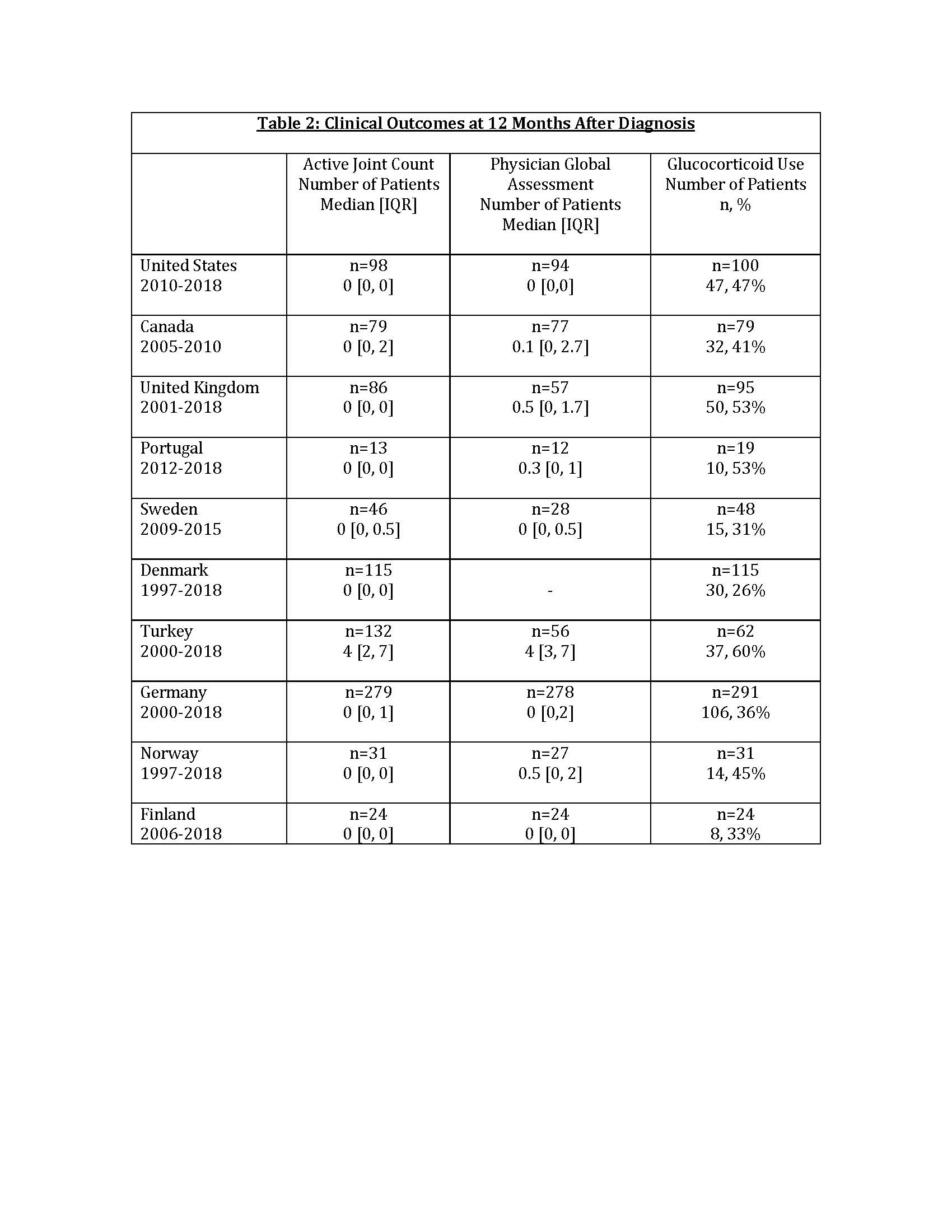
Results: A total of 1,149 patients were included for demographic data; 553 patients had medication data for the 2012-2018 era. The cohort was predominantly female and Caucasian; median age at first presentation to PR ranged from 5.3-8 years. Median duration of symptoms prior to first visit with PR varied from 0-3.3 months. GC use was common in the first year of diagnosis, with a weighted average of 72% (range 33-96%) of patients prescribed GC in the first year over the duration of each Registry’s collection. Biologic use included IL-1, IL-6 and TNF inhibitors. There was a clear increase in the proportion of patients treated with biologics, primarily anakinra, after 2012 (Table 1). Weighted mean PGA and AJC at the 12±3 month visit were 1.55 and 1.57, respectively (Table 2). At the 12±3 month visit, the proportion of patients prescribed GC varied (weighted mean 40%, range 26-60%).
Conclusion: Analysis of SJIA patients across 10 international Registries demonstrated that patients were seen by a PR at variable times from symptom onset. Although many local factors may influence treatment decisions, biologic use increased after 2011, consistent with secular trends. Anakinra was the most frequently prescribed biologic. Nearly 75% of patients were prescribed steroids within the first year of diagnosis; steroid use seemed to decrease at 12 months of treatment. More study is needed to determine long-term outcomes in SJIA patients treated with modern treatment regimens.
To cite this abstract in AMA style:
Son M, Kimura Y, Aalto K, Berntson L, Dallas J, Duffy C, Glerup M, Guzman J, Herlin T, Hovi P, Hyrich K, Klotsche J, Magnusson B, McIntyre V, Nordal E, Ozen S, Santos M, Sozeri B, Beukelman T. The Initial Treatment of Systemic Juvenile Idiopathic Arthritis: An International Collaboration Among 10 Registries [abstract]. Arthritis Rheumatol. 2020; 72 (suppl 4). https://acrabstracts.org/abstract/the-initial-treatment-of-systemic-juvenile-idiopathic-arthritis-an-international-collaboration-among-10-registries/. Accessed .« Back to 2020 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/the-initial-treatment-of-systemic-juvenile-idiopathic-arthritis-an-international-collaboration-among-10-registries/