Session Information
Session Type: ACR Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose: It is important for internists to know how to appropriately and effectively order subspecialty labs. As part of the choosing wisely campaign in 2013, the American College of Rheumatology (ACR) issued a series of recommendations to improve clinical practice including not to test ANA subserologies without a positive F-ANA.1
Multiple studies and laboratory guidelines have evaluated this algorithmic approach which begins with the clinical suspicion of a rheumatic disease and then assessment of F-ANA and ANA sub-serologies as indicated.2–6
Methods: A retrospective review of medical records of patients admitted to our large, urban medical center in an underserved area from June 2016 through June 2017 was performed. 460 patients in whom F-ANA or ANA-ENA panel testing was performed were examined. Case records were analyzed for the number of tests requested, the sequence of the testing, and test results. Indeterminate results in the ANA-ENA panel were not taken into consideration for the analysis. Data was analyzed using Excel 2013.
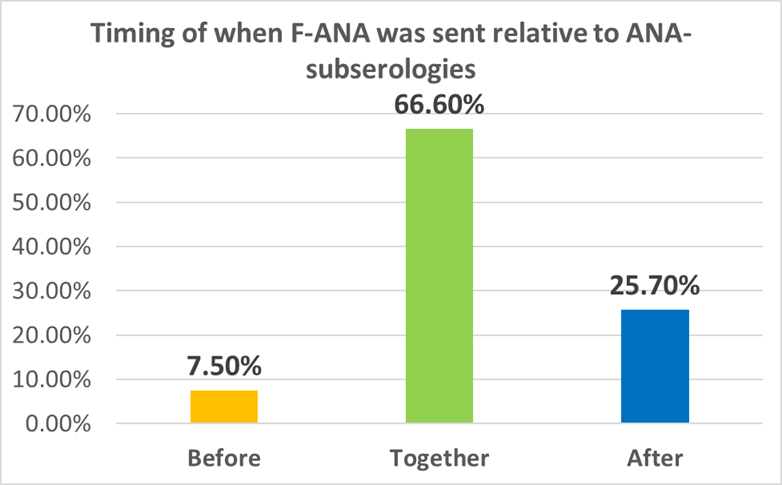
Results: One hundred ninety F-ANA and 429 ENA panels were ordered. Positive results were found in 56.3% (107/190) of F-ANA and 14.6% (63/429) of the ENA panels. When both F-ANA and ENA panels were requested, F-ANA was performed before the ENA panel as recommended in the minority of patients 7.5% (12/159). The majority had the two tests done at the same time 66.6% (106/159). 25.7% (41/159) had the F-ANA sent after the ENA panels. ENA panels were requested alone the majority of the time 62.9% (270/429). Females represented 57.6% of the patients.
Conclusion: The majority of the time a F-ANA was not ordered before the subserologies as per ACR recommendations. Therefore we conclude that in our hospital population the ACR guidelines are not being followed with the potential for decreased quality of care and missed diagnosis. This may be due to the cost constraints of the hospital where finances are limited and F-ANA costs $14.81 and ANA-ENA panel costs $6.09. Additionally, in our ordering system, it is not obvious that the ANA-ENA panel does not include the F-ANA. We suggest further education of the house staff about understanding the difference between the F-ANA and ANA-ENA panel as a point to start toward quality improvement in this area. Also, we feel that these results open the question of whether other hospitals and communities with limited resources are able to follow the ACR guidelines for F-ANA and ENA ordering due to financial constraints. Further study in this area could help further inform guidelines going forward.
To cite this abstract in AMA style:
Ramirez de Oleo IE, Johnson B, Mendez-Agrusa B. QI Project: Inpatient Practices in Ordering Fluorescent Anti-Nuclear Antibodies (F-ANA) and ANA Subserologies in a City Hospital in an Underserved Community [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 9). https://acrabstracts.org/abstract/qi-project-inpatient-practices-in-ordering-fluorescent-anti-nuclear-antibodies-f-ana-and-ana-subserologies-in-a-city-hospital-in-an-underserved-community/. Accessed .« Back to 2018 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/qi-project-inpatient-practices-in-ordering-fluorescent-anti-nuclear-antibodies-f-ana-and-ana-subserologies-in-a-city-hospital-in-an-underserved-community/