Session Information
Session Type: Poster Session (Tuesday)
Session Time: 9:00AM-11:00AM
Background/Purpose: Rheumatoid arthritis (RA) is a chronic autoimmune disorder that is associated with increased risk of cardiovascular disease, cardiometabolic disorders, and autoimmune disease. Thus, we examined the hypothesis that genetic predisposition to RA increases the risk for cardiovascular disease, cardiometabolic disorders, and other autoimmune disorders.
Methods: To test this hypothesis, we selected 102 independent SNPs (R2< 0.10) that were significantly associated with RA (P< 5×10-8) in the largest transethnic meta-analysis published. In the Vanderbilt DNA biobank (BioVU), ninety-five SNPs had a call rate ≥ 95% and were used to construct a weighted genetic risk score (wGRS) for RA to: 1) test its association with 9 cardiometabolic disorders (Table 1) and 2) perform a global phenome wide association study (PheWAS) examining the relationship between the wGRS and all phenotypes present in 29,892 Caucasian patients adjusting for median age in the electronic health record, sex, and 5 principal components. A P-value ≤ 0.006 was considered significant for the 9 selected cardiometabolic diseases and a false discovery rate (FDR) < 0.1 was used in the global PheWAS. In addition, a set of independent RA-associated SNPs (R2< 0.05) was used to perform a two-sample inverse-variance weighted regression (IVWR) meta-analysis using publicly available summary statistics for the 9 cardiometabolic phenotypes of interest (or proxies). Egger regression was performed to test horizontal pleiotropy with phenotypes in which the IVWR analysis was significant.
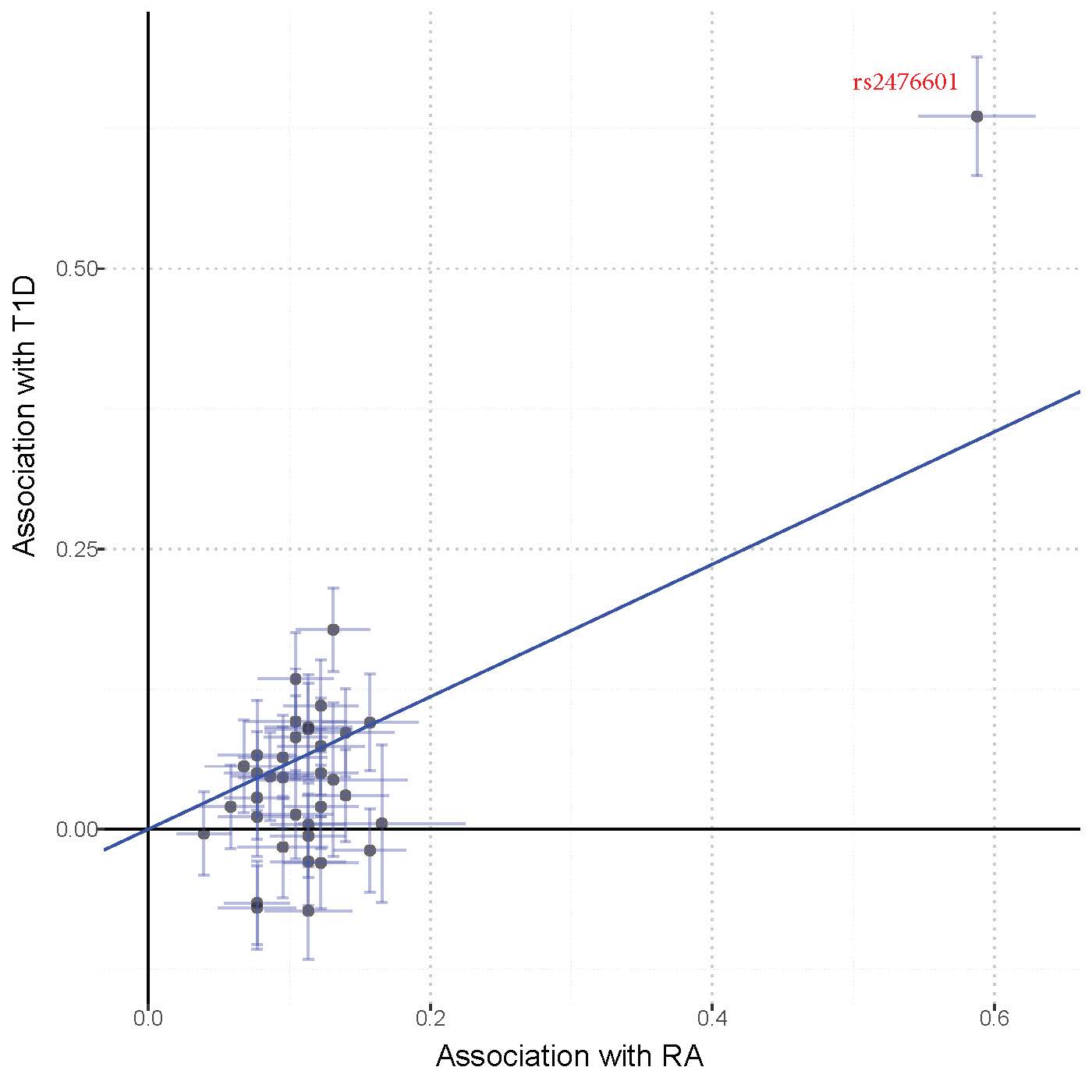
Results: Among the 9 cardiometabolic phenotypes selected, the wGRS for RA was significantly associated with type I diabetes (T1D) (P=9.82×10-4, Table 1). In the global PheWAS, the wGRS was associated with increased risk for several autoimmune phenotypes including RA (OR=1.41, P=1.54×10-27), thyroiditis, systemic sclerosis, and T1D phenotypes, and with protection against multiple sclerosis and demyelinating disorders (Fig. 1). In the IVWR analysis, RA was significantly associated with type 2 diabetes (T2D) (P=4.04×10-4, Table1), but the association was abrogated when an RA SNP in the HLA region (rs9268839) was excluded from the analysis (P=0.133). T1D was also significantly associated with RA in the IVWR analysis (P=1.93×10-10, Table 1, Fig. 2) with the Egger intercept term suggesting horizontal pleiotropy (P< 0.001). When rs2476601 (an RA SNP in PTPN22) was excluded, the IVWR analysis remained significant (P=1×10-4) and the intercept term in the Egger regression no longer showed evidence of horizontal pleiotropy (P=0.611).
Conclusion: We found evidence suggesting pleiotropy between genetic predisposition for RA and several other autoimmune disorders including T1D. The IVWR analysis showed that genetic predisposition to RA was associated with increased risk of T1D, even after the exclusion of a common variant in PTPN22 known to be associated with different autoimmune disorders.
To cite this abstract in AMA style:
Kawai V, Shi M, Feng Q, Chung C, Liu G, Cox N, Roden D, Stein C, Mosley J. Pleiotropy of Genetic Predisposition to Rheumatoid Arthritis Increases the Risk for Autoimmune Disease [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/pleiotropy-of-genetic-predisposition-to-rheumatoid-arthritis-increases-the-risk-for-autoimmune-disease/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/pleiotropy-of-genetic-predisposition-to-rheumatoid-arthritis-increases-the-risk-for-autoimmune-disease/