Session Information
Session Type: ACR Poster Session A
Session Time: 9:00AM-11:00AM
Background/Purpose:
Polymyositis (PM) and Dermatomyositis (DM) are inflammatory conditions characterized by persistent inflammation of muscle tissue (and for DM also skin). Myositis has been shown to be associated with upregulationof MMP expression and activity in muscle.
Neoepitope biomarkers are peptide biomarkers that are the product of pathology specific post-translational modification, e.g. MMP-mediated proteolytic cleavage. Several such biomarkers have shown to be modulated during inflammatory conditions such as rheumatoid arthritis and synovitis in osteoarthritis, as well as in tissue fibrosis. Thus, we hypothesized that they likewise be differentially modeulatedduring DM or PM, individually or together. Methods:
In a previously described clinical trial comprised of 61 myositis patients (28 DM, 23 PM) and 30 age and gender-matched controls, we measured the serum biomarkers MMP-cleaved collagens 1, 3, and 6 (C1M, C3M, C6M), Propeptides of collagens 1 and 3 (P1NP and PRO-C3), MMP-cleaved CPR (CRPM), MMP-cleaved titin (TIM), and caspase-cleaved Myosin light chain 3 (MYL-C3). First we analyzed biomarker levels across PM, DM and controls using one-way ANOVA. Second, we analyzed the ability of the biomarkers to separate PM and DM from healthy controls through logistic regression and Receiver-operator characteristic analysis. Third, we examined the relationship between the biomarkers and the MMT8 score (a manual measurement score of muscle function across 8 muscles), through multivariate regression analysis. Results:
We found that C1M, C3M, CRPM, PINP and PRO-C3 were differentially modulated in PM and DM relative to controls (Figure 1), in the form of downregulation of CRPM and PINP relative to controls and upregulationof PRO-C3 in PM and in C1M and C3M in DM.
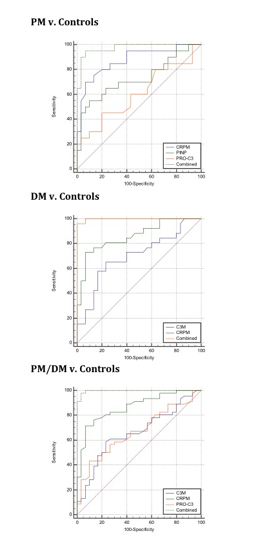
Next, we found that biomarkers panels could separate DM, PM and DM+PM from controls with 90%+ sensitivity and specificity (area under the curve for corresponding ROC curves of 0,997, 0.973 and 0,996, Figure 2).
Last, we report that biomarker panels can effectively predict MMT8 scores in PM (best model included C6M, p<0.0001), DM (best model included C6M and CRPM, p<0.0001) and DM+PM (best model included C6M, CRPM and PRO-C3, p=0.0001) Conclusion:
First, we can report that neoepitope biomarkers differentially produced in myositis patients relative to healthy controls. Second, we found that neoepitope biomarker panels can discriminate between healthy controls and DM, PM or DM/PM combined with extremely high sensitivity and specificity. Third, we report that biomarker panels, particularly C6M, closely followmuscle function.
Figure 1
Figure 2
To cite this abstract in AMA style:
Nedergaard A, Henriksen K, Karsdal MA, White W, Guo X. Neoepitope Biomarkers As Biomarkers of Polymyositis and Dermatomyositis and Functional Status [abstract]. Arthritis Rheumatol. 2015; 67 (suppl 10). https://acrabstracts.org/abstract/neoepitope-biomarkers-as-biomarkers-of-polymyositis-and-dermatomyositis-and-functional-status/. Accessed .« Back to 2015 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/neoepitope-biomarkers-as-biomarkers-of-polymyositis-and-dermatomyositis-and-functional-status/