Session Information
Date: Monday, November 11, 2019
Title: Metabolic & Crystal Arthropathies Poster II: Clinical Trials & Basic Science
Session Type: Poster Session (Monday)
Session Time: 9:00AM-11:00AM
Background/Purpose: Gout is caused by the deposition of monosodium urate (MSU) crystals in joints due to chronic hyperuricemia. Long term treatment focuses on reducing sUA levels, thus allowing MSU crystals to dissolve.
Pegylated uricases are therapies for treatment of severe chronic gout. However, uricases are limited by induction of anti-drug antibodies (ADA) that can compromise efficacy and safety. SEL-212 is a novel combination product consisting of pegadricase co-administered with proprietary ImmTOR tolerogenic nanoparticles. ImmTOR has been designed to mitigate the formation of ADAs by inducing tolerogenic dendritic cells and antigen-specific regulatory T cells. Prolonged therapeutic activity of uricase (pegadricase) is vital in maintaining a sustained control of serum uric acid (SUA) levels in patients. We report data on ADA [anti-uricase] and uricase activity from an open label study of monthly SEL-212 treatment in symptomatic gout patients.
Methods: Patients with symptomatic gout (≥1 tophus, gout flare within 6 months or gouty arthropathy) and elevated SUA ≥6 mg/dL were treated with fixed doses of pegadricase (0.2 mg/kg) in combination with ImmTOR (0.1 and 0.15 mg/kg), or pegadricase (0.2 or 0.4 mg/kg) alone as a control. Patients were infused in 28-day cycles up to 5 times. Safety, tolerability, sUA, ADAs, and uricase activity were monitored.
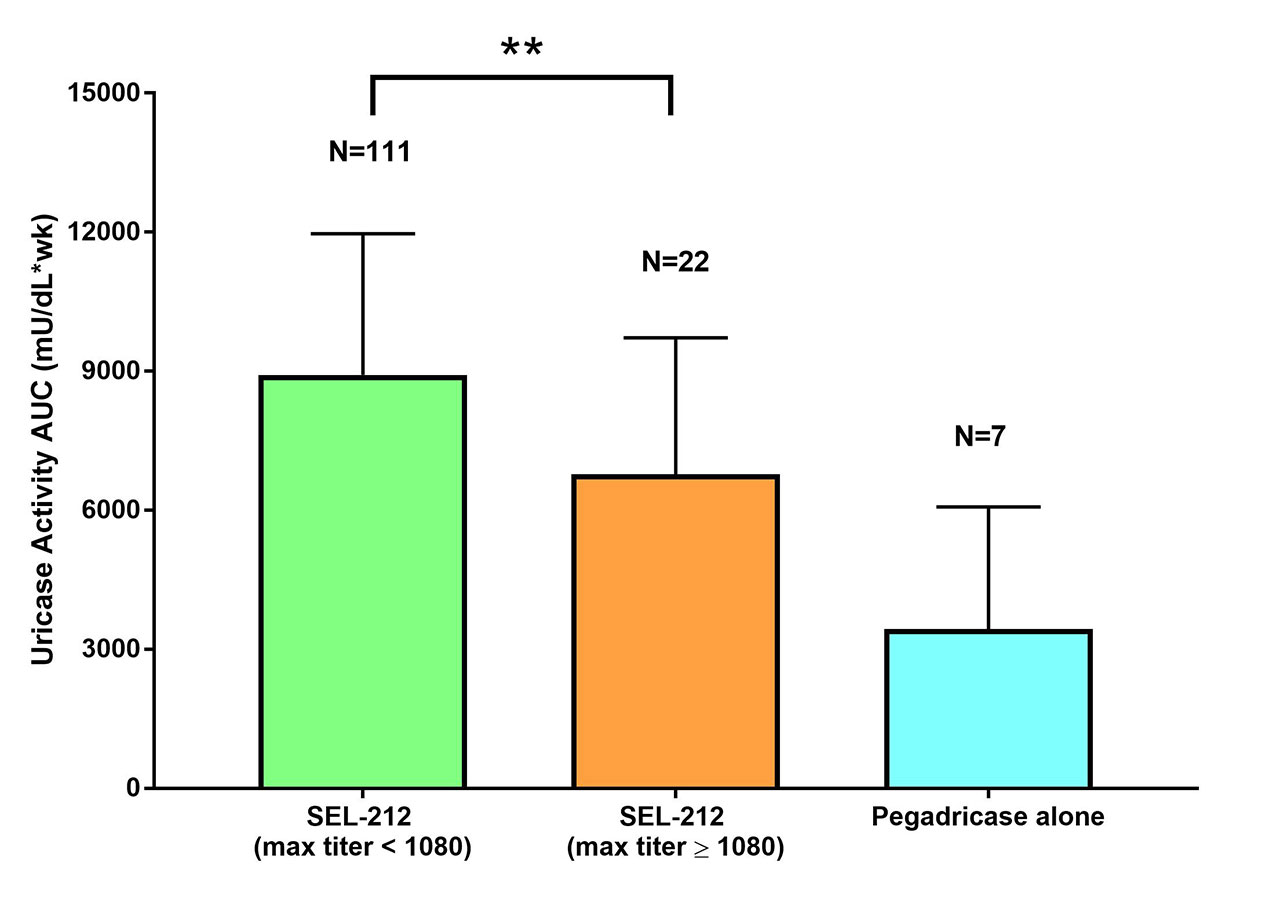
Results: Demographics of 46 patients treated with 28-day cycles x5 combination doses of ImmTOR and pegadricase as compared to 6 patients treated with pegadricase alone was 23-70 years old vs. 41-64 years old (mean 53.6 vs. 51.8 years), male 97.8% vs. 100%, and white 73.9%. vs. 33.3%. The mean BMI at baseline was 34.5 vs. 38.9 kg/m2. 71.7% vs. 100% of patients were obese with mean duration of established or symptomatic gout as 12.5 vs. 12.8 years. 43 SEL-212 patients were evaluable for this analysis. The majority of treatment periods with SEL-212 had maximum antibody titers < 1080 (83.4%, 111/133) while 14.3% (1/7) treatment periods of pegadricase alone had maximum antibody titers < 1080. AUC of uricase activity was significantly higher in treatment periods where maximum ADA titers were below 1080 (p=0.0029). For all treatment periods for patients receiving SEL-212 in which their maximum anti-uricase titer was < 1080 (n=111), the mean uricase activity AUC was 8924 ± 3044 (mean ± SD) while in treatment periods where the maximum anti-uricase titer was ≥1080 (n=22), the mean uricase activity AUC was 6776 ± 2949. In treatment periods where control cohort patients received pegadricase alone (n=7), uricase activity had a mean AUC of 3437 ± 2635 which was lower than both SEL-212 treated groups. Six of the 7 treatment periods had maximum antibody titers ≥1080. For SEL-212 patients that had titers < 1080, the uricase activity AUCs were sustained over the 5 treatment periods and corresponding serum uric acid levels remained low.
Conclusion: By mitigating the formation of ADAs, monthly dosing of ImmTOR combined with pegadricase increases uricase activity in symptomatic gout patients relative to pegadricase alone. When anti-uricase titers are < 1080, patients show sustained uricase activity, enabling 28 day treatment intervals.
To cite this abstract in AMA style:
DeHaan W, Kivitz A, Azeem R, Plotkin H, Johnston L, Kishimoto T, Park J, Smolinski S, Leung S. Monthly Dosing of ImmTOR Tolerogenic Nanoparticles Combined with Pegylated Uricase (Pegadricase) Mitigates Formation of Anti-Drug Antibodies Resulting in Sustained Uricase Activity in Symptomatic Gout Patients [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/monthly-dosing-of-immtor-tolerogenic-nanoparticles-combined-with-pegylated-uricase-pegadricase-mitigates-formation-of-anti-drug-antibodies-resulting-in-sustained-uricase-activity-in-symptomatic-gout/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/monthly-dosing-of-immtor-tolerogenic-nanoparticles-combined-with-pegylated-uricase-pegadricase-mitigates-formation-of-anti-drug-antibodies-resulting-in-sustained-uricase-activity-in-symptomatic-gout/