Session Information
Date: Monday, October 22, 2018
Title: Metabolic and Crystal Arthropathies – Basic and Clinical Science Poster I
Session Type: ACR Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose:
Pegylated uricases are therapies for treatment of severe chronic gout, particularly for rapid tophi resolution. However, uricases are limited by induction of anti-drug antibodies (ADA) that can compromise efficacy and safety. SEL-212 is a novel combination product consisting of pegadricase (also known as pegsiticase) co-administered with synthetic vaccine particles encapsulating rapamycin (SVP-R). We report initial data on gout flares from an ongoing Phase 2 study in symptomatic gout patients.
Gout is caused by deposition of monosodium urate (MSU) crystals in joints due to chronic hyperuricemia. Long term treatment focuses on reducing sUA levels, allowing MSU crystals to dissolve. Rapid dissolution of MSU crystals during initial phase of urate lowering therapy (ULT) is associated with an increased frequency of acute gout flares, which can contribute to poor treatment compliance. During ULT initiation, colchicine, NSAIDs or corticosteroids are used for gout flare prophylaxis.
Methods:
Patients with symptomatic gout (≥1 tophus, gout flare within 6 months or gouty arthropathy) and elevated serum uric acid (sUA) ≥6 mg/dL were treated with fixed doses of pegadricase (0.2 mg/kg or 0.4 mg/kg) alone or in combination with SVP-Rapamycin (0.05 to 0.15 mg/kg). SEL-212 was infused in 28-day cycles x3 doses followed by challenge with pegadricase alone on 28-day cycles x2 doses, or in 28-day cycles x5 combination doses of SVP-Rapamycin and pegadricase. Safety, tolerability, sUA, and ADAs were monitored.
All randomized patients received colchicine (1.2 mg as loading dose, 0.6 mg QD for the remainder of their participation in the trial) as premedication for gout flare prevention. If colchicine was contraindicated, patients received ibuprofen 600 mg TID or equivalent dose of a NSAID. If colchicine and NSAIDs were contraindicated, patients did not receive any premedication.
Results:
As of 21 May 2018, demographics of the 140 treated patients were 32 – 75 years old (mean 54.9 years), male 90.7%, and white 67.1%. The mean BMI at baseline was 35.0 kg/m2. 70.0% of patients were obese with mean duration of established or symptomatic gout as 10.8 years.
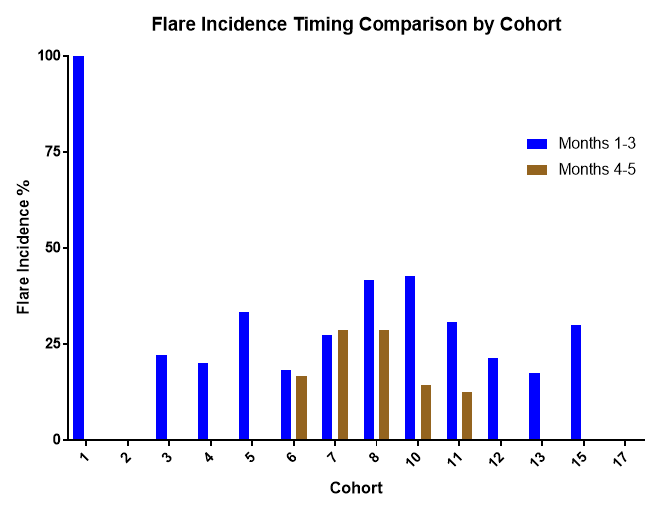
Flare incidence was 27.1% (months 1-3) and 10.2% (months 4-5), flare frequency was 0.41 flares/patient (months 1-3) and 0.14 flares/patient (months 4-5). Mean duration of the gout flares was 7.28 days, with majority of the gout flares (93.8%) being categorized as mild/moderate, with 6.1% (n=4 cases) noted as severe in intensity. Adjustments to gout flare prevention medication were not required for 43% of the patients. No gout flares resulted in a patient discontinuation or were reported as a serious adverse event.
Figure 1: Comparison of Gout Flare Incidence
Conclusion:
SEL-212 has been well-tolerated and lowers flares at the initiation of therapy relative to pegylated uricases alone, and the effect persists over the duration of therapy.
To cite this abstract in AMA style:
Azeem R, Kivitz AJ, Sands E, DeHaan Ph.D. W, Johnston L, Kishimoto TK. Initial Phase 2 Clinical Data of SEL-212 in Symptomatic Gout Patients: Monthly Dosing of a Pegylated Uricase (Pegadricase) with Svp-Rapamycin Enables Sustained Reduction of Acute Gout Flares [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 9). https://acrabstracts.org/abstract/initial-phase-2-clinical-data-of-sel-212-in-symptomatic-gout-patients-monthly-dosing-of-a-pegylated-uricase-pegadricase-with-svp-rapamycin-enables-sustained-reduction-of-acute-gout-flares/. Accessed .« Back to 2018 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/initial-phase-2-clinical-data-of-sel-212-in-symptomatic-gout-patients-monthly-dosing-of-a-pegylated-uricase-pegadricase-with-svp-rapamycin-enables-sustained-reduction-of-acute-gout-flares/