Session Information
Date: Sunday, November 8, 2020
Title: RA – Treatments Poster III: PROs, Biomarkers, Systemic Inflammation & Radiographs
Session Type: Poster Session C
Session Time: 9:00AM-11:00AM
Background/Purpose: In the FINCH 1 study, filgotinib (FIL)—an oral, potent, selective JAK1 inhibitor—plus methotrexate (MTX) provided significant improvements in signs and symptoms of rheumatoid arthritis (RA) in patients (pts) with inadequate response to MTX.1 EULAR guidelines recommend a treat-to-target approach focused on reducing inflammation to prevent joint damage, physical disability, and mortality, but pts consider pain and fatigue control, and maintenance of physical function and health-related quality of life (HRQoL), to be important aspects of care.2,3 Here, we present the rate and magnitude of change in PROs from FINCH 1.
Methods: In FINCH 1 (NCT02889796), pts with active RA received oral FIL 200 mg + MTX, FIL 100 mg + MTX, PBO + MTX, or subcutaneous adalimumab (ADA) 40 mg + MTX for up to 52 weeks (W); pts receiving PBO at W24 were rerandomized 1:1 to FIL 100 or 200 mg.
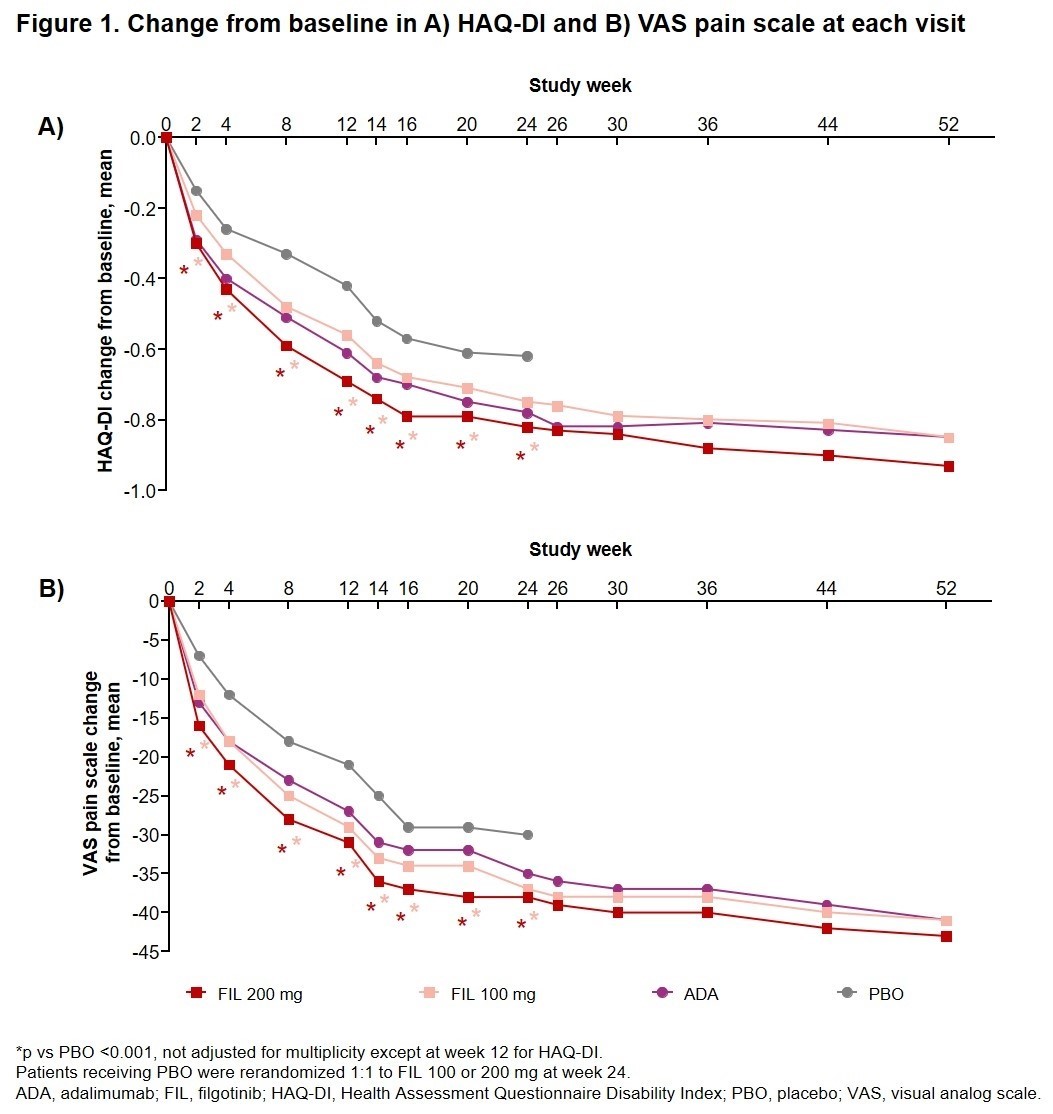
PROs included the HAQ-DI and VAS pain scale, SF-36, and FACIT-Fatigue questionnaire. The change from baseline (CFB) at each time point was assessed up to W52 for each treatment group. Mixed-effects model for repeated measures was used to compare each FIL group with PBO for the CFB at each time point through W24. logistic regression model was used to compare each FIL group with PBO for the proportion of pts achieving the minimum clinically important difference (MCID) of ≥0.22 reduction in CFB in HAQ-DI at each time point through W24.
Results: Of 1755 pts randomized and treated (475 FIL 200 mg + MTX; 480 FIL 100 mg + MTX; 325 ADA + MTX; and 475 PBO + MTX), 1417 (80.7%) received study drug through W52. As early as W2 through W24, pts receiving either FIL dose experienced nominally significantly greater (p < 0.001) CFB in HAQ-DI and VAS pain scale than those receiving PBO; CFB improvements were maintained through W52 (Fig 1A, B). At W2, compared with PBO (40.2%), a nominally significantly greater proportion of pts achieved the HAQ-DI MCID in both the FIL 200 (52.5%; p < 0.001) and 100 mg (46.7%; p = 0.043) groups. This benefit vs PBO was maintained up to W24 and the proportion of pts who achieved a HAQ-DI reduction ≥0.22 remained ≥75.8% in the FIL 200 mg group and ≥71.5% in the FIL 100 mg group from W12 through W52. FIL provided nominally significantly greater improvement in HRQoL vs PBO at W4 and W12 for both the CFB of the SF-36 Physical Component Summary (PCS; p < 0.001) and Mental Component Summary (MCS;p ≤0.006); nominal significance was also seen at W24 for CFB of SF-36 PCS (Fig 2A, B). By W4, pts receiving either dose of FIL reported a nominally significantly greater mean CFB in FACIT-Fatigue scores vs PBO (p < 0.001); significance was maintained through W24 and improvement in reported fatigue continued through W52 in the FIL groups (Fig 2C). In general, CFB for HAQ-DI, VAS pain scale, and FACIT-Fatigue observed for the FIL groups was higher or comparable to ADA at various time points (Fig 1, 2).
Conclusion: Both doses of FIL provided rapid and sustained improvements in functional status, pain, HRQoL, and fatigue vs PBO for pts with RA and inadequate response to MTX throughout the 52-week period.
References:
- Combe BG, et al. Ann Rheum Dis. 2019;78 (Suppl 2):A77.
- Fautrel B, et al. Rheumatol Int. 2018;38:935–47.
- Smolen JS, et al. Ann Rheum Dis. 2017;76:960–77.
To cite this abstract in AMA style:
Kivitz A, Tanaka Y, Lee S, Ye L, Hu H, Besuyen R, Combe B. Filgotinib Provided Rapid and Sustained Improvements in Functional Status, Pain, Health-related Quality of Life, and Fatigue in Patients with Rheumatoid Arthritis and Inadequate Response to Methotrexate [abstract]. Arthritis Rheumatol. 2020; 72 (suppl 10). https://acrabstracts.org/abstract/filgotinib-provided-rapid-and-sustained-improvements-in-functional-status-pain-health-related-quality-of-life-and-fatigue-in-patients-with-rheumatoid-arthritis-and-inadequate-response-to-methotrexa/. Accessed .« Back to ACR Convergence 2020
ACR Meeting Abstracts - https://acrabstracts.org/abstract/filgotinib-provided-rapid-and-sustained-improvements-in-functional-status-pain-health-related-quality-of-life-and-fatigue-in-patients-with-rheumatoid-arthritis-and-inadequate-response-to-methotrexa/