Session Information
Date: Saturday, November 12, 2022
Title: RA – Treatment Poster I
Session Type: Poster Session A
Session Time: 1:00PM-3:00PM
Background/Purpose: The safety profile of filgotinib (FIL), a second-generation oral Janus kinase (JAK) 1 preferential inhibitor approved in Europe, Japan, and the UK for treatment of RA,1,2 has been reported.3 In patients (pts) with active RA aged ≥50 y with ≥1 cardiovascular (CV) risk factor, treated with the pan-JAK inhibitor tofacitinib, data from an interventional post-marketing study (NCT02092467)4 suggested a higher risk of major adverse cardiovascular events (MACE) and malignancies compared with TNF inhibitors. No data are available from a similar prospective study with FIL. This post hoc exploratory analysis aimed to describe the incidence of MACE and malignancies in a subgroup of pts with RA from the FINCH and DARWIN clinical trials, receiving FIL 200 mg (FIL200) and FIL 100 mg (FIL100).
Methods: Exploratory analysis of adverse events of special interest are reported using integrated FIL RA data from phase 2 (NCT01668641, NCT01894516), phase 3 (NCT02889796, NCT02873936, NCT02886728), and the long-term extension (LTE) studies DARWIN 3 phase 2 (NCT02065700) and FINCH 4 phase 3 (NCT03025308), in a pt population at higher risk of CV events similar to ORAL-SURVEILLANCE,1 namely, aged ≥50 y with ≥1 CV risk factor (history of dyslipidemia, diabetes or CV disease; hypertension, ischemic vascular conditions, peripheral vascular disease, extra-articular manifestations of RA; or current smokers). All pts met ACR criteria for functional class I–III. Censored exposure-adjusted incidence rates (EAIRs)/100 pt-years of exposure for MACE, venous thromboembolism (VTE), malignancies excluding nonmelanoma skin cancer (NMSC), NMSC, (serious) infections, and deaths were determined. Data were as of Jan 11, 2022 (DARWIN 3) and Jan 31, 2022 (FINCH 4). Analyses were performed on an ad hoc interim analysis data set without additional cleaning.
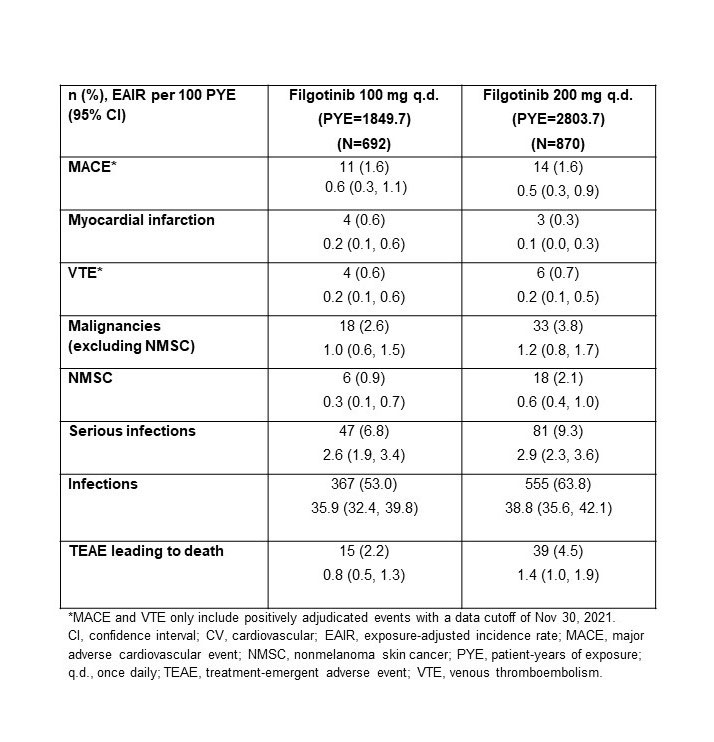
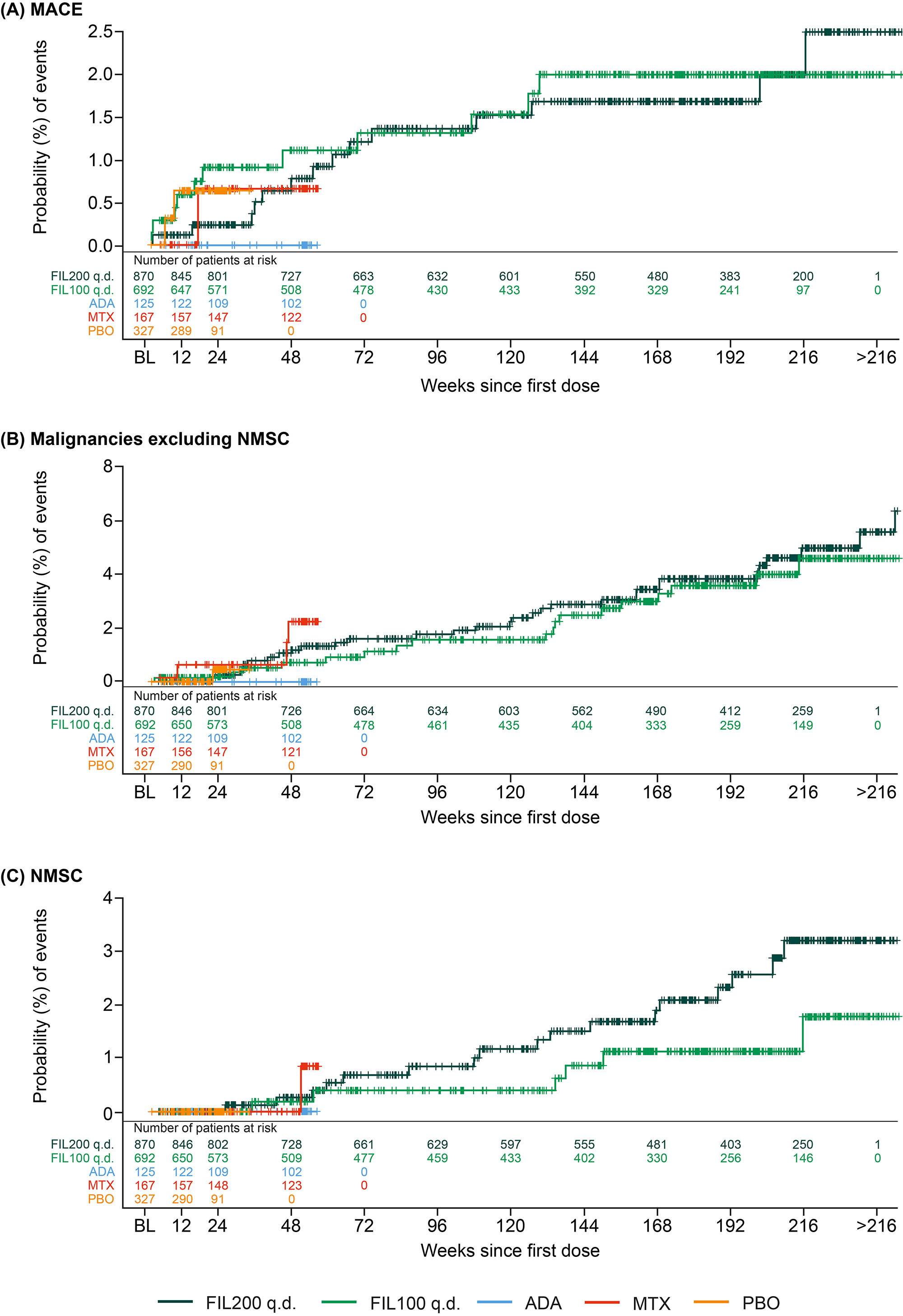
Results: The higher-risk population included 1484 pts: mean age 62.1 y (536 [36.1%] aged ≥65 y), 291 (22.5%) current smokers, and 904 (60.9%) received background MTX; baseline (BL) MTX use was higher in the FIL100 vs FIL200 group (66.6% vs 54.6%); other BL demographics were balanced. Numerically higher EAIRs for malignancies (excluding NMSC), NMSC, serious infections, and deaths were observed for pts receiving FIL200 vs FIL100; 95% confidence intervals overlapped (Table). For MACE and VTE, incidence is considered similar for both doses (Table). The risk of experiencing MACE, malignancies excluding NMSC, and NMSC over time with FIL100 or FIL200 is shown in the Figure.
Conclusion: In this RA population at higher risk of CV events, the incidence of MACE and VTE was similar for both doses. A numerically higher incidence of malignancies (excluding NMSC), NMSC, serious infections, and deaths was observed in the high- vs low-dose group. Limitations included population selection bias, low event numbers, and the post hoc nature of the analysis. The ongoing LTE and real-world studies in RA will continue to investigate the safety of FIL in these populations.
References
1. Jyseleca SmPC. Galapagos NV; May 2022
2. Jyseleca Japanese PI. Gilead Sciences K.K.; Sep 2020
3. Winthrop KL, et al. Ann Rheum Dis 2022;81:184–92
4. Ytterberg SR, et al. N Engl J Med 2022;386:316–26
To cite this abstract in AMA style:
Buch M, Burmester G, Mariette X, Charles-Schoeman C, Rajendran V, Stiers P, Cerani A, Van Hoek P, Van Beneden K, Tanaka Y, Schulze-Koops H, Westhovens R, Favalli E. Exploratory Analysis of Filgotinib Safety Data in Patients with Moderately to Severely Active RA and an Increased Risk of Cardiovascular Events: Data from Phase 2 and 3 Clinical Trials [abstract]. Arthritis Rheumatol. 2022; 74 (suppl 9). https://acrabstracts.org/abstract/exploratory-analysis-of-filgotinib-safety-data-in-patients-with-moderately-to-severely-active-ra-and-an-increased-risk-of-cardiovascular-events-data-from-phase-2-and-3-clinical-trials/. Accessed .« Back to ACR Convergence 2022
ACR Meeting Abstracts - https://acrabstracts.org/abstract/exploratory-analysis-of-filgotinib-safety-data-in-patients-with-moderately-to-severely-active-ra-and-an-increased-risk-of-cardiovascular-events-data-from-phase-2-and-3-clinical-trials/