Session Information
Date: Sunday, November 7, 2021
Title: Metabolic & Crystal Arthropathies – Basic & Clinical Science Poster I (0660–0682)
Session Type: Poster Session B
Session Time: 8:30AM-10:30AM
Background/Purpose: Eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), also known as omega-3 fats act as alternate COX substrates to arachidonic acid (AA). Their metabolism results in production of prostaglandins which are less inflammatory than those derived from AA. Thus omega-3 fatty acids can act as a “natural” anti-inflammatory drug. Regular use of anti-inflammatory doses of omega 3 fish oil may be particularly useful as a prophylaxis against gout flares during the introduction of urate lowering therapy, and acceptable to patients due to tolerability and lack of major adverse effects compared to other prophylactic medications. The aim of this study was determine the effect of omega-three supplementation with fish oil on serum urate (SU), weight and body mass index (BMI) in people with gout.
Methods: A pilot 6-month, randomized, open-label clinical trial was undertaken in people with gout with serum urate ≥6mg/dL/l. Forty participants were randomized to receive 6.2g omega-3 fish oil daily (manufactured by Melrose (Mt Waverley, Victoria, Australia)or no fish oil for 24 weeks. Blood was obtained monthly for SU and red cell EPA (20:5n-3) DHA (22:6n-3) were measured using a blood spot collection system. The primary efficacy outcome was the change in SU between baseline and week 24 (or the final visit for those who discontinued fish oil or were lost to follow-up). Secondary outcomes included (i) change in weight and BMI between baseline and week 24, (ii) mean number of flares per month between baseline and week 24, (iii) percentage of participants with flares at each month, (iv) cessation of fish oil therapy prior to week 24 due to adverse effects or intolerance and (v) change in oxypurinol, HBA1c, cholesterol, HDL, and LDL concentrations between baseline and week 24 for people on allopurinol.
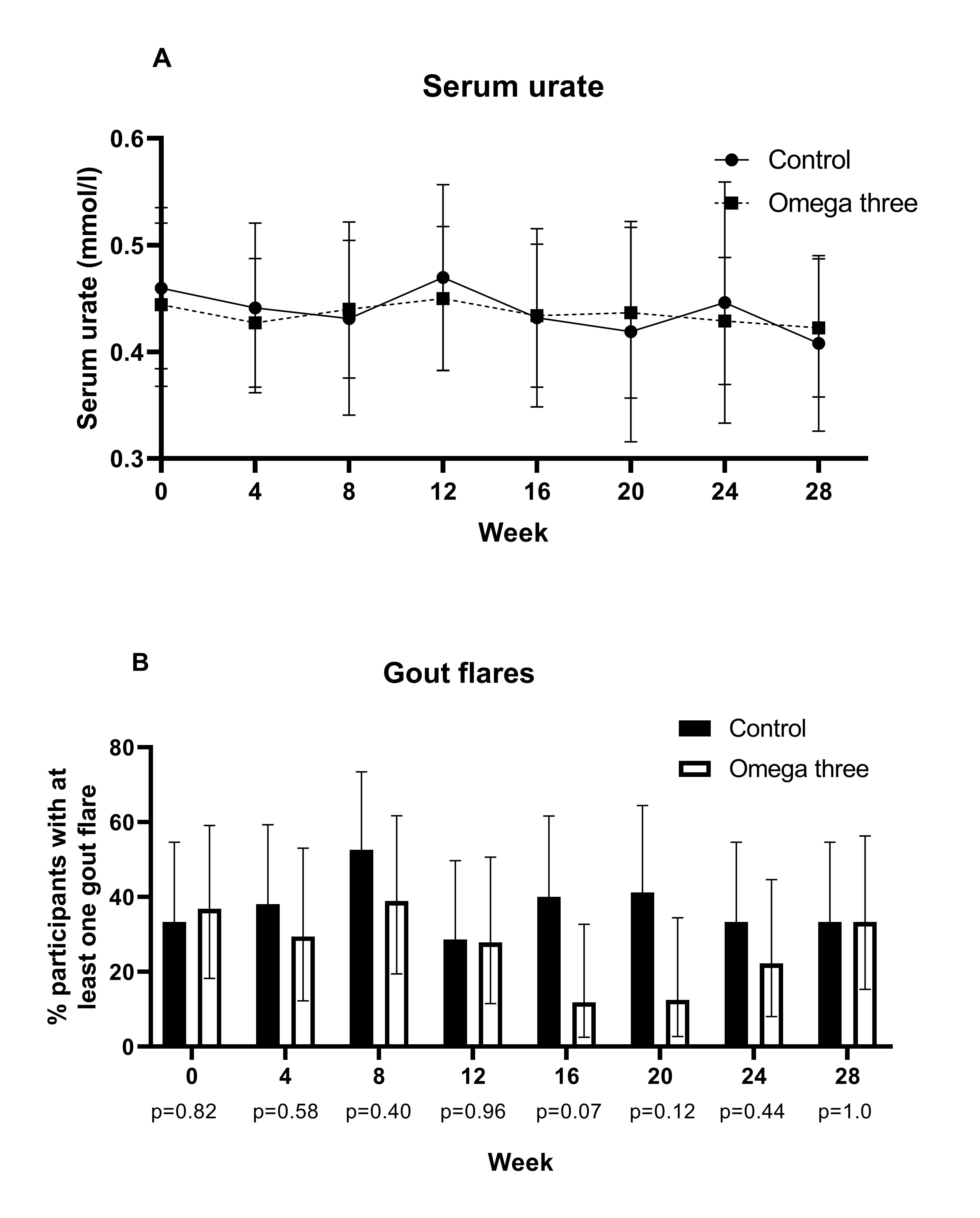
Results: Five participants in the fish oil group discontinued the oil; three were unable to tolerate the taste, one commenced dabigatran and one found it inconvenient to take it. The baseline demographic and clinical features were well matched between randomized groups. There was no statistically significant difference in the mean (SEM) decrease in SU between baseline and week 24 between randomized groups: fish oil -0.021 (0.02) mmol/l vs. control -0.006 (0.02) mmol/l. There was no significant difference in change in weight or BMI between baseline and week 24 between randomized groups (Figure). There was a statistically significant correlation between red cell omega-three concentrations and the total number of flares per participant between week 12 and week 24; total omega-three r= -0.75 (p≤0.001), EPA r= -0.75 (p≤ 0.001) and DHA r= -0.76 (p≤ 0.001). In the omega-three fish oil group four participants reported gastrointestinal adverse effects definitely or probably related to the omega-three supplementation.
Conclusion: The lack of untoward effect of omega three fish oil supplementation on SU and BMI together with the relationship between higher omega-three concentrations and lower gout flares supports the development of further adequately powered clinical trials to determine the role of omega-three supplements as prophylaxis against gout flares in people starting urate lowering therapy.
To cite this abstract in AMA style:
Stamp L, Grainger R, Frampton C, Drake J, Hill C. Effect of Omega-three Supplementation on Serum Urate and Gout Flares in People with Gout; A Pilot Randomized Trial [abstract]. Arthritis Rheumatol. 2021; 73 (suppl 9). https://acrabstracts.org/abstract/effect-of-omega-three-supplementation-on-serum-urate-and-gout-flares-in-people-with-gout-a-pilot-randomized-trial/. Accessed .« Back to ACR Convergence 2021
ACR Meeting Abstracts - https://acrabstracts.org/abstract/effect-of-omega-three-supplementation-on-serum-urate-and-gout-flares-in-people-with-gout-a-pilot-randomized-trial/