Session Information
Session Type: Poster Session (Monday)
Session Time: 9:00AM-11:00AM
Background/Purpose: Use of abatacept, a targeted DMARD (tDMARD), in patients with RA has shown to improve whole-body insulin sensitivity and reduce HbA1c levels.1 Comparative evidence on type 2 diabetes mellitus (T2DM) related outcomes against other tDMARDs is lacking. We measured the healthcare resource utilization (HCRU) and costs associated with T2DM in tDMARD-naïve Medicare beneficiaries with RA and T2DM who initiated treatment with abatacept, tumor necrosis factor inhibitors (TNFi), or other non-TNFi.
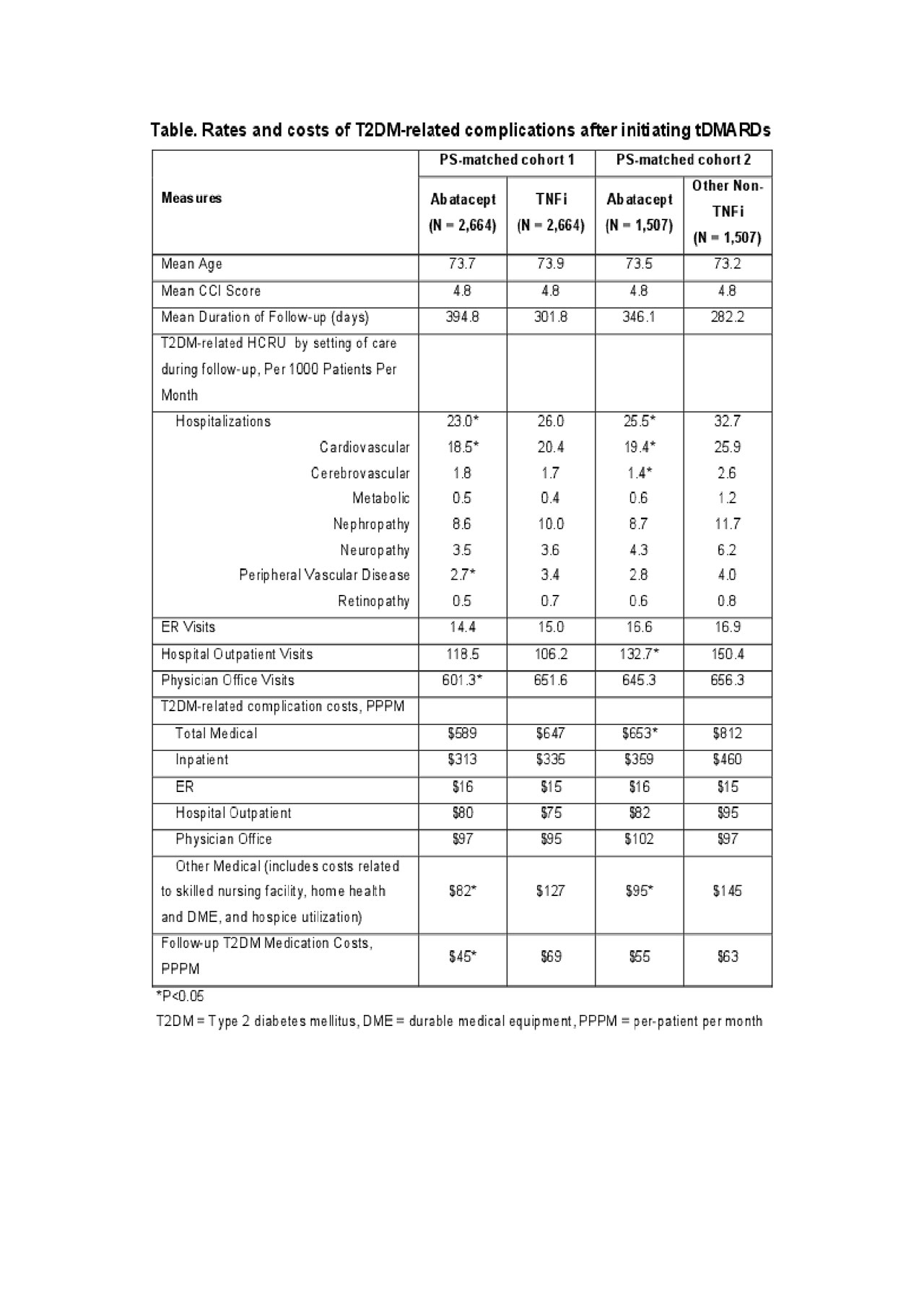
Methods: Patients initiating abatacept, a TNFi (adalimumab, certolizumab pegol, etanercept, golimumab, infliximab), or other non-TNFi (anakinra, rituximab, sarilumab, tocilizumab, tofacitinib) from 2010 through 2017 were identified in a retrospective cohort using 100% Medicare fee-for-service (FFS) claims (Parts A/B/D). Elderly patients (≥65 years of age) were included if they had ≥2 diagnoses of RA, ≥1 diagnosis of T2DM or treated with T2DM medications, no recent history of cancer, and were continuously enrolled for 12 months pre-index date or the date of tDMARD initiation. Post-index follow-up was until discontinuation of the index treatment, disenrollment, death, or end of study period, whichever occurred first. Patients who initiated abatacept were propensity score (PS) matched to initiators of TNFi and non-TNFi separately on baseline demographics, comorbidities, medications, T2DM-related healthcare resource use and costs. T2DM-related complications include neuropathy, nephropathy, cerebrovascular, cardiovascular, retinopathy, peripheral vascular disease and metabolic from the validated Diabetes Severity Complication Index (DCSI).2 The HCRU of T2DM-related complications was measured using the counts of inpatient stay, ER visits and outpatient visits per 1,000 patients per month whereas costs (2019 USD) were measured as per-patient per month (PPPM).
Results: A total of 2,664 PS-matched pairs of abatacept and TNFi’s users, and 1,507 PS-matched pairs of abatacept and other non-TNFi’s users were identified. During follow-up, the rate of hospitalizations associated with T2DM-related complications per 1,000 patients per month was the lowest in abatacept group compared to both TNFi (23.0 vs. 26.0) and other non-TNFi groups (25.5 vs. 32.7). Similar trends were observed for ER visits and outpatient care. The major driver of T2DM-related healthcare costs for each group was hospitalizations. PPPM T2DM-related complication costs decreased following tDMARD initiation in abatacept (˃20%) and TNFi (17%) users and increased in non-TNFi users (1.5%). During follow-up, PPPM T2DM-related costs were $58 and $159 lower for abatacept versus TNFi and non-TNFi initiators, respectively.
Conclusion: In Medicare FFS beneficiaries with RA and T2DM, abatacept initiators had lower T2DM-related HCRU and costs compared to initiators of TNFis and non-TNFis. These findings suggest that use of abatacept as a first line tDMARD could help reduce the clinical and economic burden associated with T2DM in patients with RA.
References:
- Ursini F, et al. Medicine (Baltimore). 2015;94(21):e888.
- Glasheen WP, et al. J Diabetes Complications. 2017;31(6):1007-1013.
To cite this abstract in AMA style:
Patel V, Pulungan Z, Shah A, Jones B, Petrilla A, Ferri L, Han X, Michaud K. Comparison of Healthcare Resource Utilization and Costs of Type 2 Diabetes Mellitus (T2DM)-Related Complications in Medicare Beneficiaries with Rheumatoid Arthritis (RA) and T2DM Who Initiated Treatment with Abatacept versus Other Targeted Disease-Modifying Anti-Rheumatic Drugs [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/comparison-of-healthcare-resource-utilization-and-costs-of-type-2-diabetes-mellitus-t2dm-related-complications-in-medicare-beneficiaries-with-rheumatoid-arthritis-ra-and-t2dm-who-initiated-treatme/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/comparison-of-healthcare-resource-utilization-and-costs-of-type-2-diabetes-mellitus-t2dm-related-complications-in-medicare-beneficiaries-with-rheumatoid-arthritis-ra-and-t2dm-who-initiated-treatme/