Session Information
Date: Saturday, November 6, 2021
Title: Abstracts: Spondyloarthritis Including PsA – Treatment I: Emerging Therapies (0488–0491)
Session Type: Abstract Session
Session Time: 2:45PM-3:00PM
Background/Purpose: Bimekizumab (BKZ), a monoclonal IgG1 antibody that selectively inhibits both interleukin (IL)-17F and IL-17A, has been demonstrated to be efficacious and well tolerated in patients (pts) with ankylosing spondylitis (AS) treated for up to 156 weeks (wks).1–3 We report 3-year (yr) interim safety and efficacy of BKZ in pts with active AS from a 1-yr phase 2b study (BE AGILE; NCT02963506) and its ongoing 4-yr open-label extension (OLE; NCT03355573).
Methods: The BE AGILE study design has been described previously.1 Following the 12-wk double-blind, placebo-controlled dose-ranging period, pts received BKZ 160 mg or 320 mg every 4 wks (Q4W) in the dose-blind period to Wk 48. Pts completing Wk 48 were eligible to enter the OLE where all pts received BKZ 160 mg Q4W. We report results following a total of 3 yrs of treatment. Treatment-emergent adverse events (TEAEs) are reported separately for BKZ exposure in BE AGILE and the OLE (safety sets). Efficacy outcomes are reported primarily for the dose-blind set. Analyses utilized non-responder imputation (NRI) and observed case (OC) methodology, or multiple imputation (MI); for NRI, pts who did not enter the OLE were considered non-responders from Wk 48 onwards.
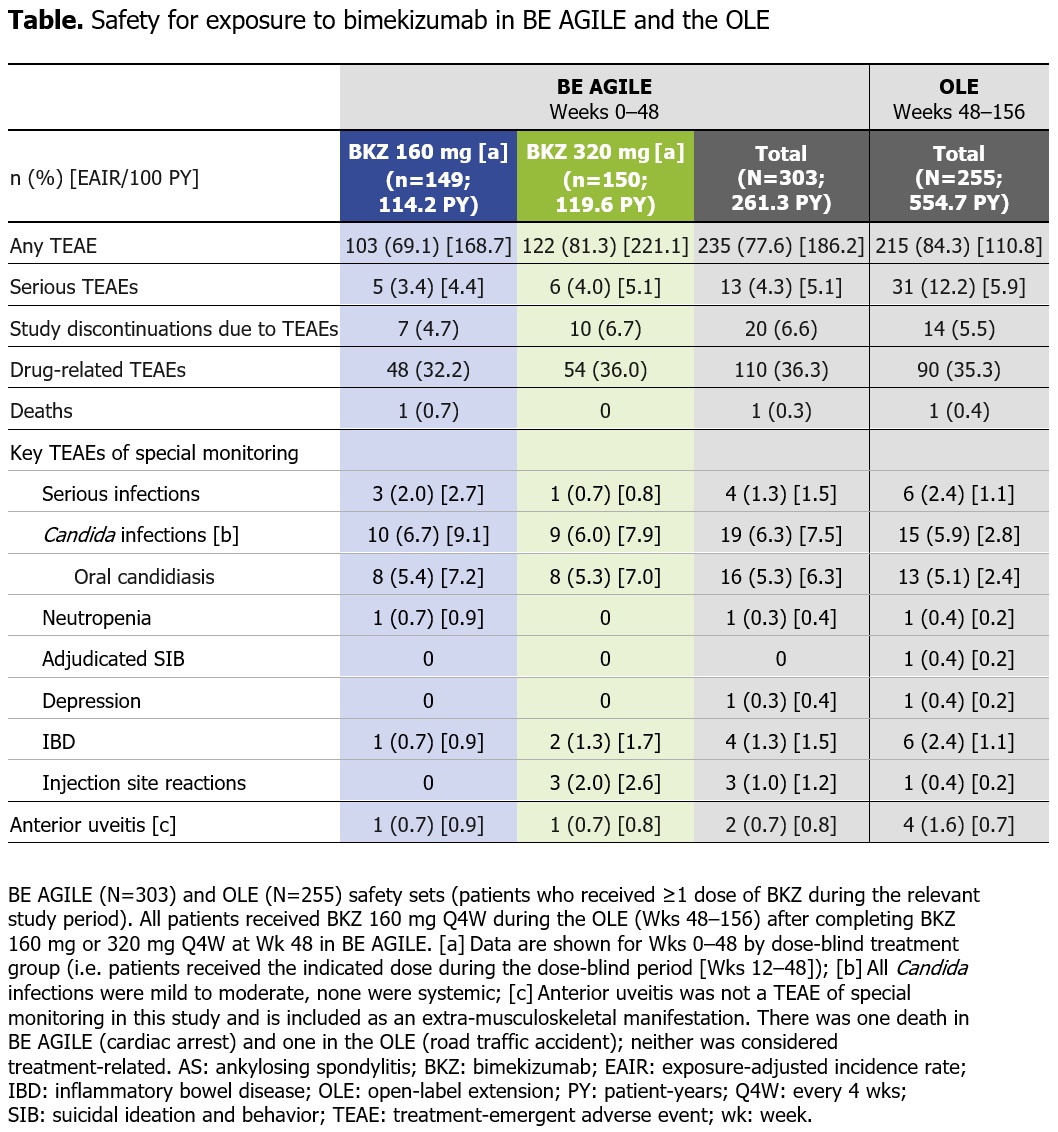
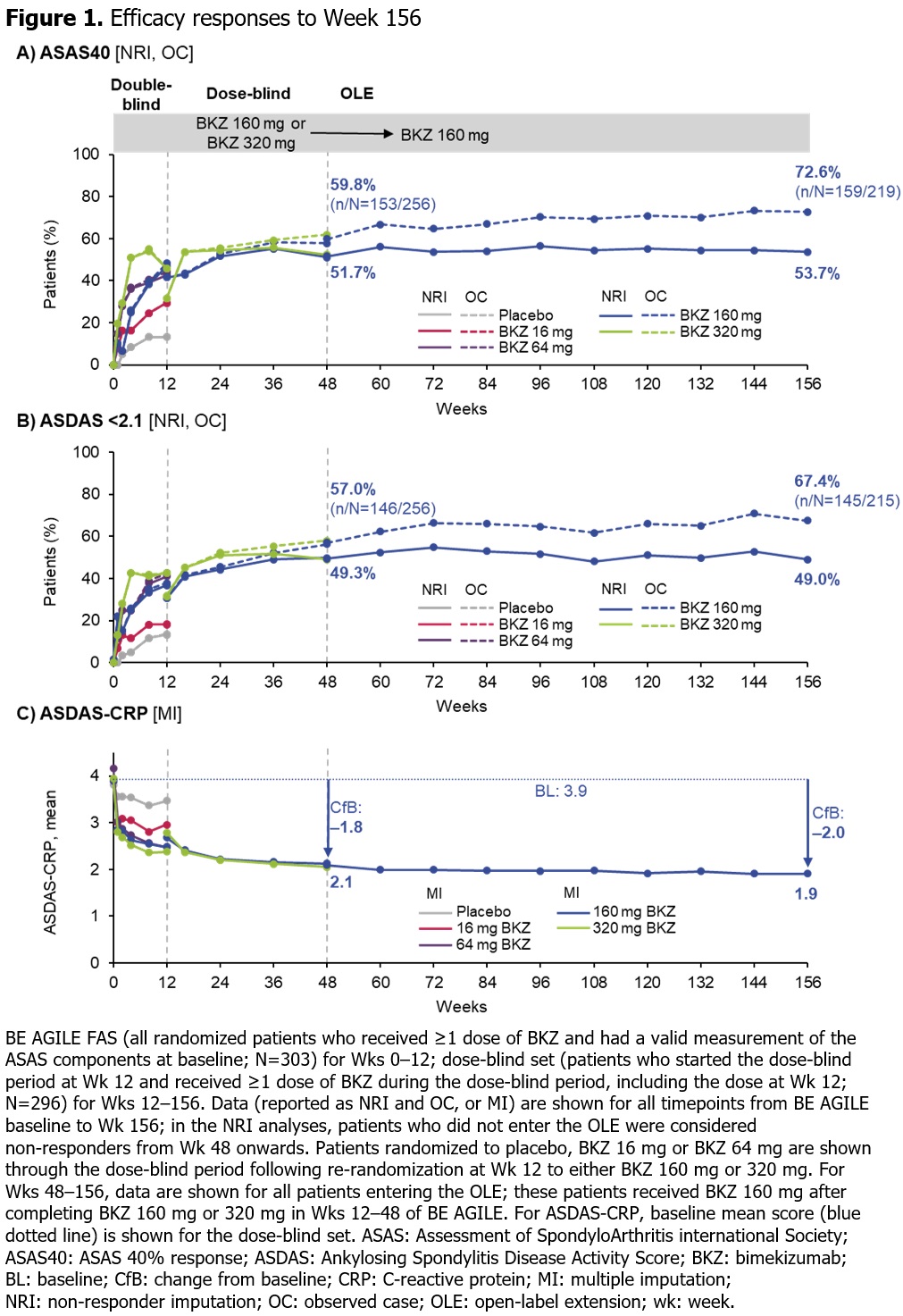
Results: 296/303 (97.7%) pts randomized at BE AGILE study baseline (BL) entered the dose-blind period at Wk 12; 262/303 (86.5%) pts completed Wk 48 on BKZ. At Wk 48, 256/303 (84.5%) pts entered the OLE, of whom 255 received BKZ 160 mg Q4W; 224/303 (73.9%) pts completed Wk 156. For Wks 0–48 (and the OLE), exposure-adjusted incidence rates (EAIRs) per 100 pt‑yrs were 186.2 (OLE: 110.8) for any TEAE, 5.1 (OLE: 5.9) for serious TEAEs, 1.5 (OLE: 1.1) for serious infections, and 7.5 (OLE: 2.8) for Candida infections (Table). Overall, EAIRs of TEAEs in the OLE remained stable or were lower than in Wks 0–48. The majority of Candida infections were oral candidiasis; all Candida infections were mild or moderate. EAIRs of inflammatory bowel disease, anterior uveitis, and injection site reactions were low across BE AGILE and the OLE (Table). In the NRI analyses, the percentage of pts achieving ASAS40 response or ASDAS < 2.1 was sustained from Wk 48 to Wk 156 (Figure 1A–B). Following substantial improvements from BL to Wk 48 in mean ASDAS‑CRP (3.9 to 2.1) and BASDAI total score (6.5 to 3.0), responses were sustained over a further 2 yrs of BKZ treatment to 1.9 and 2.5, respectively, at Wk 156 (Figure 1D & 2A). All BASDAI components demonstrated continued improvements in pt‑reported fatigue, spinal pain, joint pain and swelling, discomfort due to tenderness to touch or pressure, and intensity and duration of morning stiffness (Figure 2B).
Conclusion: The long-term safety profile of BKZ in pts with AS was in line with previous observations, with no new safety signals identified after 3-yr BKZ exposure.1–3 Efficacy was maintained and consistent over 3 yrs of BKZ treatment, highlighting the potential of dual inhibition of IL-17F and IL-17A in the treatment of AS.
References: 1. van der Heijde D. Ann Rheum Dis 2020;79:595–604; 2. Baraliakos X. Arthritis Rheumatol 2020;72 (suppl 10):1364;3. van der Heijde D. Ann Rheum Dis 2021;80 (suppl 1):332–3.
To cite this abstract in AMA style:
Gensler L, Deodhar A, van der Heijde D, Poddubnyy D, Kivitz A, Dougados M, de Peyrecave N, Oortgiesen M, Vaux T, Fleurinck C, Baraliakos X. Bimekizumab Long-Term Safety and Efficacy in Patients with Ankylosing Spondylitis: Interim Results After 3 Years of Treatment in an Ongoing Phase 2b Study [abstract]. Arthritis Rheumatol. 2021; 73 (suppl 9). https://acrabstracts.org/abstract/bimekizumab-long-term-safety-and-efficacy-in-patients-with-ankylosing-spondylitis-interim-results-after-3-years-of-treatment-in-an-ongoing-phase-2b-study/. Accessed .« Back to ACR Convergence 2021
ACR Meeting Abstracts - https://acrabstracts.org/abstract/bimekizumab-long-term-safety-and-efficacy-in-patients-with-ankylosing-spondylitis-interim-results-after-3-years-of-treatment-in-an-ongoing-phase-2b-study/