Session Information
Date: Monday, October 22, 2018
Title: 4M102 ACR Abstract: Misc Rheum & Inflam DZ I: DADA2, Cardiac Sarcoid,Cancer Immunotherapy(1911–1916)
Session Type: ACR Concurrent Abstract Session
Session Time: 4:30PM-6:00PM
Background/Purpose: The scenario of biological-triggered autoimmune diseases has dramatically changed in recent years due to the increasing use of biologics in patients with solid cancers.
Methods: In 2006, the SEMI Spanish Study Group on Autoimmune Diseases (GEAS) created the BIOGEAS project, including the BIOGEAS systematic review of the literature (articles published until January 1st 2018) using the terms of the main groups of cancer immunotherapies that were crossed with autoimmune diseases using MedDRA terms (MedDRAVR 15.0). The autoimmune basis of the reported adverse event was defined according to the fulfilment of the corresponding international criteria, biopsy-proven histopathology, positive autoantibodies and/or resolution after corticosteroid treatment.
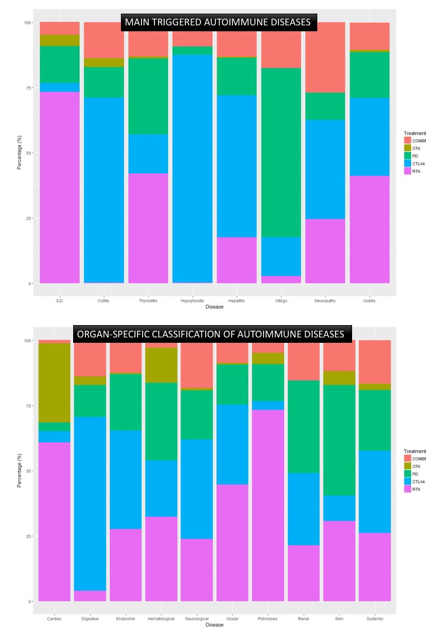
Results: We identified 6158 patients who developed 6288 autoimmune adverse events in patients with solid (n=5849) and haematological (n=309) neoplasia. The two main groups of cancer immunotherapies involved included checkpoint inhibitors in 3844 (mainly the CTL4 inhibitor ipilimumab in 1973 cases and the PD1 inhibitors pembrolizumab and nivolumab in 712 and 700 cases, respectively) and tyrosine kinase (TK) inhibitors in 2443 cases (mainly erlotinib and imatinib in 499 and 437 cases, respectively). Organ-specific classification of triggered autoimmune diseases included endocrine (n=1723), digestive (n=1591), pulmonary (n=1337), cutaneous (n=531), systemic/rheumatic (n=331), cardiac (n=304), neurological (n=218), ocular (n=150), haematological (n=38) and renal (n=69) autoimmune diseases. Figure shows distribution of the main pharmacological groups according to the most frequently-reported individual autoimmune diseases and to their classification by organ/system. The main organ-specific patterns of association (> 50% of cases related to a specific pharmacological pathway) are interstitial lung disease in patients treated with drugs targeting TK receptor; colitis, hepatitis and hypophysitis in those treated with CTL4 inhibitors; and vitiligo in those treated with drugs targeting the PD/PD-1 pathway.
Conclusion: More than 6000 cases of autoimmune diseases triggered by cancer immunotherapies have been reported. A multidisciplinary approach is essential, with a central role for the specialist in autoimmune diseases who should get used to managing not only spontaneous autoimmune diseases, but also the increasing number of biological-induced autoimmune diseases that may be forecast.
To cite this abstract in AMA style:
Retamozo S, Flores-Chavez A, Kostov B, Sisó-Almirall A, Pérez-de-Lis M, Pérez-Alvarez R, Brito-Zerón P, Khamashta MA, Ramos-Casals M. Big Data Analysis of Autoimmune Diseases Induced By Cancer Immunotherapies: Identifying Pharmacological Patterns of Organ-Specific Involvement [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 9). https://acrabstracts.org/abstract/big-data-analysis-of-autoimmune-diseases-induced-by-cancer-immunotherapies-identifying-pharmacological-patterns-of-organ-specific-involvement/. Accessed .« Back to 2018 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/big-data-analysis-of-autoimmune-diseases-induced-by-cancer-immunotherapies-identifying-pharmacological-patterns-of-organ-specific-involvement/