Session Information
Session Type: Poster Session C
Session Time: 9:00AM-11:00AM
Background/Purpose: Interstitial lung disease (ILD) causes significant complications and mortality in patients with rheumatoid arthritis (RA). We have previously shown that patients with RA-ILD had increased levels of serum IgA and IgM antibodies against human serum albumin modified with malondialdehyde-acetaldehyde (MAA) in addition to having higher levels of ACPA. Moreover, lung tissues from RA-ILD patients demonstrated increased levels of MAA antigen that co-localized with citrullinated (CIT) proteins, Type II Collagen (CII) and Vimentin (VIM), suggesting that MAA may act as a cofactor that increases the immunogenicity of CIT proteins in RA-ILD patients. Therefore, antibody responses to dually modified (MAA and CIT) VIM or CII were evaluated as biomarker(s) to differentiate RA-ILD from RA and idiopathic pulmonary fibrosis (IPF), a condition with marked histopathologic similarities to RA-ILD.
Methods: Serum was collected from patients with RA, RA-ILD, or IPF (n=15 for each group). All patients with RA fulfilled the 1987 ACR classification criteria. The presence of RA-ILD was confirmed by a board-certified subspecialist (pulmonologist or rheumatologist) and the presence of supportive chest computed tomography (CT) findings. Patients were defined as having RA without ILD in the absence of a clinical diagnosis of ILD or past chest imaging findings suggestive of ILD. All patients with IPF without underlying autoimmune disease were diagnosed by a board-certified pulmonologist and had confirmatory findings of usual interstitial pneumonia (UIP) by chest CT imaging. Samples were assessed for CCP and relative units of IgG, IgM, and IgA antibody levels to CII-MAA-CIT and VIM-MAA-CIT via ELISA. Data was analyzed using a one-way ANOVA with Tukey’s multiple comparison test.
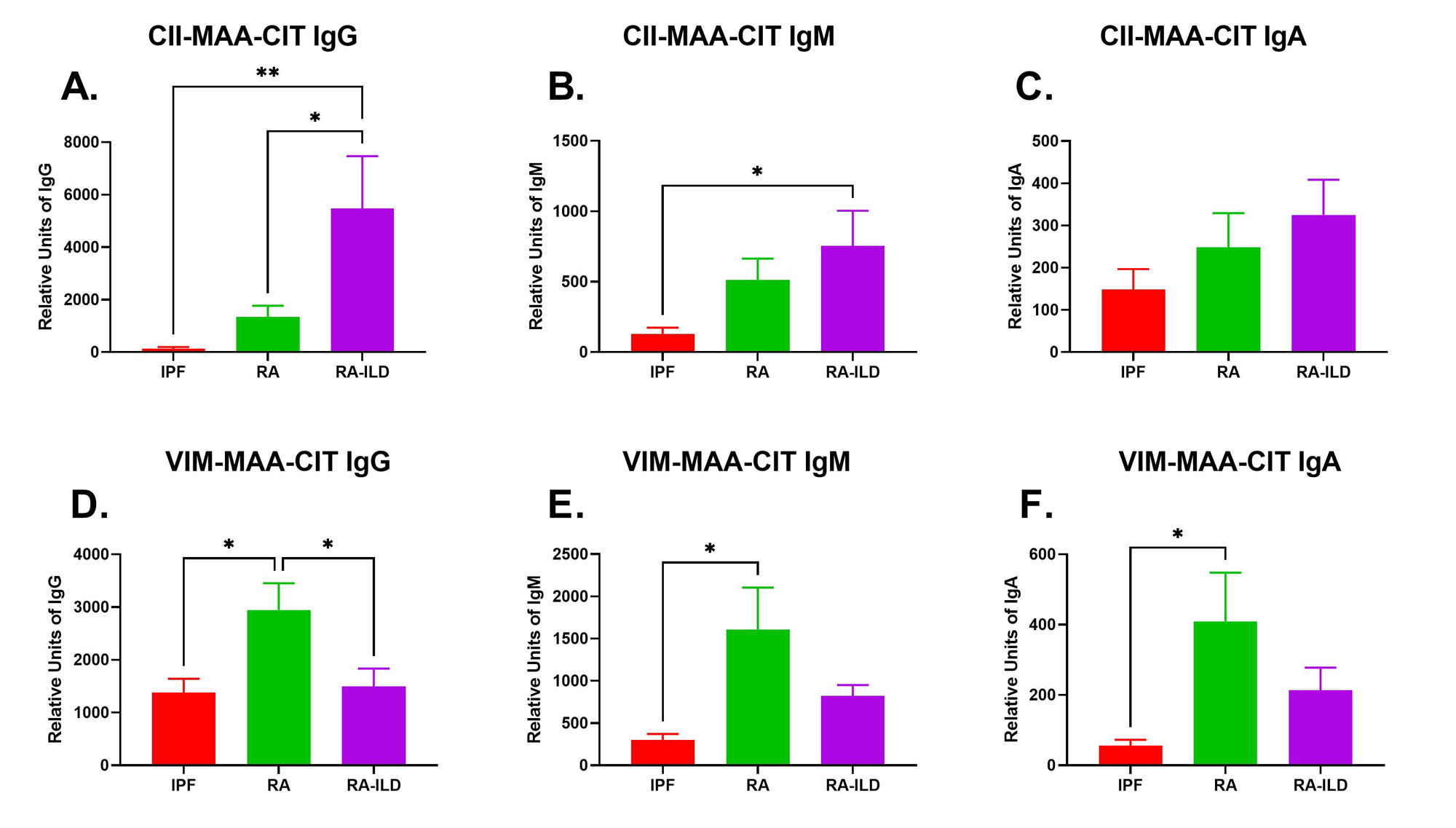
Results: Participant characteristics were similar across groups: overall mean age 73 years, 90% male, 87% reporting White race and 68.9% current or former smokers. There was no difference in CCP positivity between RA and RA-ILD patients. Serum IgG antibodies to CII-MAA-CIT were significantly increased in RA-ILD patients compared to RA patients (4-fold higher, p˂0.05) and IPF patients (42.5-fold higher, p˂0.01) (Fig. 1A). Serum IgM (Fig. 1B) and IgA (Fig. 1C) antibodies to CII-MAA-CIT showed similar trends with IgG anti-CII-MAA-CIT, but only differences in RA-ILD vs. IPF reached statistical significance (p˂0.05). In contrast, serum IgG antibodies to VIM-MAA-CIT were significantly increased in patients with RA patients when compared to those with RA-ILD (2-fold higher, p˂0.05) and 2 IPF patients (2.1-fold higher, p˂0.05) (Fig. 1D).Serum IgM (Fig. 1E) and IgA (Fig. 1F) antibodies to VIM-MAA-CIT showed a similar pattern to IgG anti-VIM-MAA-CIT, but again only differences between RA and IPF reached significance (p˂0.05).
Conclusion: Serum IgG antibody levels to dually-modified CII-MAA-CIT and VIM-MAA-CIT differ between patients with RA and RA-ILD. These findings suggest that dual modifications of type II collagen, but not vimentin, generate autoimmune responses that are substantially enhanced in RA-ILD and that could play a role in the pathogenesis of this extra-articular complication.
To cite this abstract in AMA style:
Butler B, Poole J, Duryee M, Aripova N, Hunter C, Kramer B, O'Dell J, Thiele G, England B, Mikuls T. Antibody Responses to Citrullinated Type II Collagen and Vimentin Modified with Malondialdehyde-Acetaldehyde Differ in Rheumatoid Arthritis and Rheumatoid Arthritis-Interstitial Lung Disease [abstract]. Arthritis Rheumatol. 2023; 75 (suppl 9). https://acrabstracts.org/abstract/antibody-responses-to-citrullinated-type-ii-collagen-and-vimentin-modified-with-malondialdehyde-acetaldehyde-differ-in-rheumatoid-arthritis-and-rheumatoid-arthritis-interstitial-lung-disease/. Accessed .« Back to ACR Convergence 2023
ACR Meeting Abstracts - https://acrabstracts.org/abstract/antibody-responses-to-citrullinated-type-ii-collagen-and-vimentin-modified-with-malondialdehyde-acetaldehyde-differ-in-rheumatoid-arthritis-and-rheumatoid-arthritis-interstitial-lung-disease/