Session Information
The 2020 Pediatric Rheumatology Symposium, originally scheduled for April 29 – May 2, was postponed due to COVID-19; therefore, abstracts were not presented as scheduled.
Date: Saturday, May 2, 2020
Title: Poster Session 3
Session Type: ACR Abstract Session
Session Time: 4:15PM-5:15PM
Background/Purpose: Despite being a well-established biomarker for classification of aggressive bony disease in adults with RA, ACPA have not yet been described in the ILAR criteria for polyarticular JIA. The primary aim of this project was to utilize the Childhood Arthritis and Rheumatology Research Alliance (CARRA) registry to study whether ACPA measured in the serum of patients with polyarticular JIA are associated with increased risk of bony erosions as seen on musculoskeletal imaging at the time of presentation. A secondary aim was to report the sensitivity and specificity of ACPA for erosive bony disease as compared to RF.
Methods: The eligibility criteria for inclusion in the analysis cohort were: 1) definition of polyarticular JIA met (more than four active joints at presentation), 2) musculoskeletal imaging assessing erosive disease available at presentation, 3) at least one serum ACPA and RF value available at or after presentation. Logistic regression was performed to estimate the effect of ACPA (binary independent variable) on erosions (binary dependent variable). We also calculated if adjusting for age at presentation, gender and duration of symptoms changes the effect estimate of ACPA for erosive disease. To estimate sensitivity and specificity, two-by-two tables were prepared for ACPA and RF with erosive disease. For analysis of proportions, Chi Square test was employed and a p-value of less than 0.05 was considered statistically significant. Cohen’s kappa statistic (scale 0-1) was used to measure the agreement between ACPA and RF. All data analysis was performed in RStudio version 1.1.456. The study protocol underwent exempt review by the joint Institutional Review Board at Tufts Medical Center and Boston Children’s Hospital.
Results:
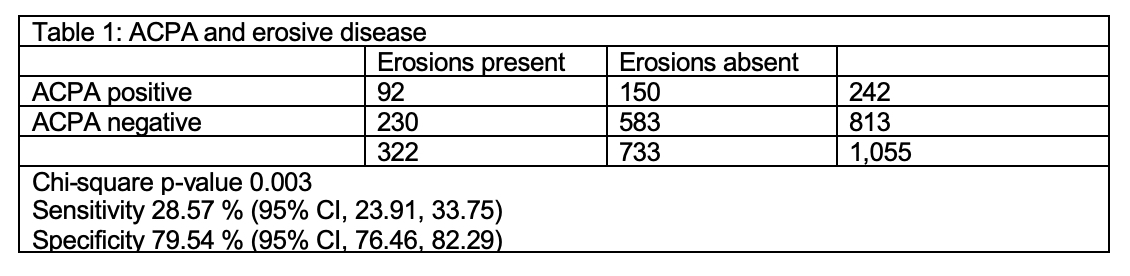
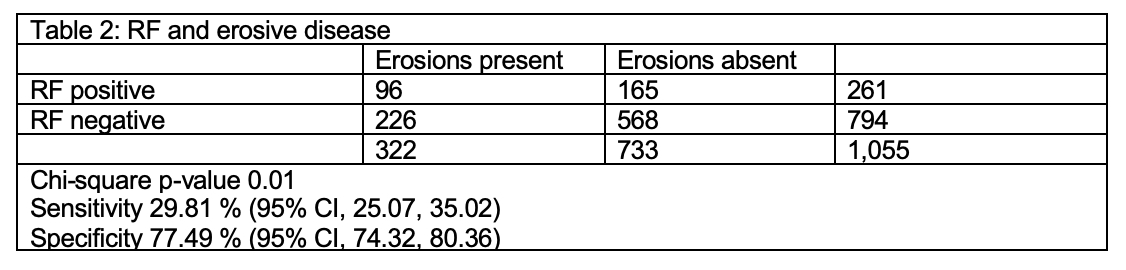
A total of 1,055 patients met the eligibility criteria in the registry from June 2015 to January 2018. In this cohort 79% were female and 74% white with a mean duration of joint symptoms of 9.6 months (SD 15.5). About 31% (322) patients had erosive disease at the time of presentation. Out of the 322 patients with erosive disease, 82 had both RF and ACPA positive, 216 had both negative, 10 patients had ACPA positive but RF negative and another 14 had ACPA negative but RF positive. The crude Odds Ratio (OR) for ACPA and erosive disease was 1.55 (95% CI 1.14, 2.09). Adjusting for gender, age at presentation and duration of symptoms did not change the effect estimate of ACPA (adjusted OR 1.54, 95% CI 1.12, 2.11). The crude OR for RF and erosive disease was 1.46 (95% CI 1.08, 1.96). ACPA had a specificity of 79.5% and a sensitivity of 28.6% for erosive disease (Table 1). RF had specificity of 77.5% and sensitivity of 29.8% (Table 2). Combining these tests modestly increased the sensitivity to 32.9% if either was positive and increased specificity to 83.2% if both were positive (Table 3). There was substantial agreement between ACPA and RF (Cohen’s kappa statistic 0.75, 95% CI 0.71, 0.80).
Conclusion:
ACPA was found to be associated with erosive bony disease and performed comparably to RF in this cohort of early polyarticular JIA patients. These findings might support the inclusion of ACPA in the diagnostic criteria for polyarticular JIA in any future revisions of ILAR criteria.
To cite this abstract in AMA style:
Joshi S, Zhang Y, Davis T. Anti-Citrullinated Protein Antibodies (ACPA) and Bony Erosions in Polyarticular JIA [abstract]. Arthritis Rheumatol. 2020; 72 (suppl 4). https://acrabstracts.org/abstract/anti-citrullinated-protein-antibodies-acpa-and-bony-erosions-in-polyarticular-jia/. Accessed .« Back to 2020 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/anti-citrullinated-protein-antibodies-acpa-and-bony-erosions-in-polyarticular-jia/