Session Information
Date: Monday, November 11, 2019
Title: 4M115: Pain Mechanisms – Basic & Clinical Science (1878–1883)
Session Type: ACR Abstract Session
Session Time: 4:30PM-6:00PM
Background/Purpose: Many patients with RA report persistent pain in the absence of clinically appreciable inflammation. In a mouse model of inflammatory arthritis, mice injected with anti-cyclic citrullinated peptide (CCP) antibody exhibited increased pain sensitivity and more pain behaviors in the absence of histological evidence of joint inflammation. These findings raise the possibility of an inflammation-independent pain pathway mediated by anti-CCP antibodies. In this study, we investigated the association between anti-CCP antibody and pain, assessed by quantitative sensory testing (QST) and patient-reported measures, in RA patients with active inflammation.
Methods: This cross-sectional analysis included study participants who met the ACR/EULAR 2010 classification criteria for RA, had active inflammatory disease, and required initiation of a disease-modifying antirheumatic drug (DMARD). Serum levels of anti-CCP antibody and high sensitivity C-reactive protein (hsCRP) were measured. Subjects also underwent QST, including assessment of pressure pain thresholds (PPTs) at wrist and knee joints to quantify peripheral sensitization. Self-reported pain measures were obtained and included average pain intensity on a numeric rating scale (NRS) and pain interference score assessed by the Patient Reported Outcomes Measurement Information System computerized adaptive test (PROMIS® CAT). We examined the cross-sectional relation of anti-CCP antibody seropositivity to PPT and self-reported pain measures using linear regression, adjusting for potential confounders. We also assessed for a dose-response relationship by categorizing anti-CCP as seronegative and as quintiles of seropositivity.
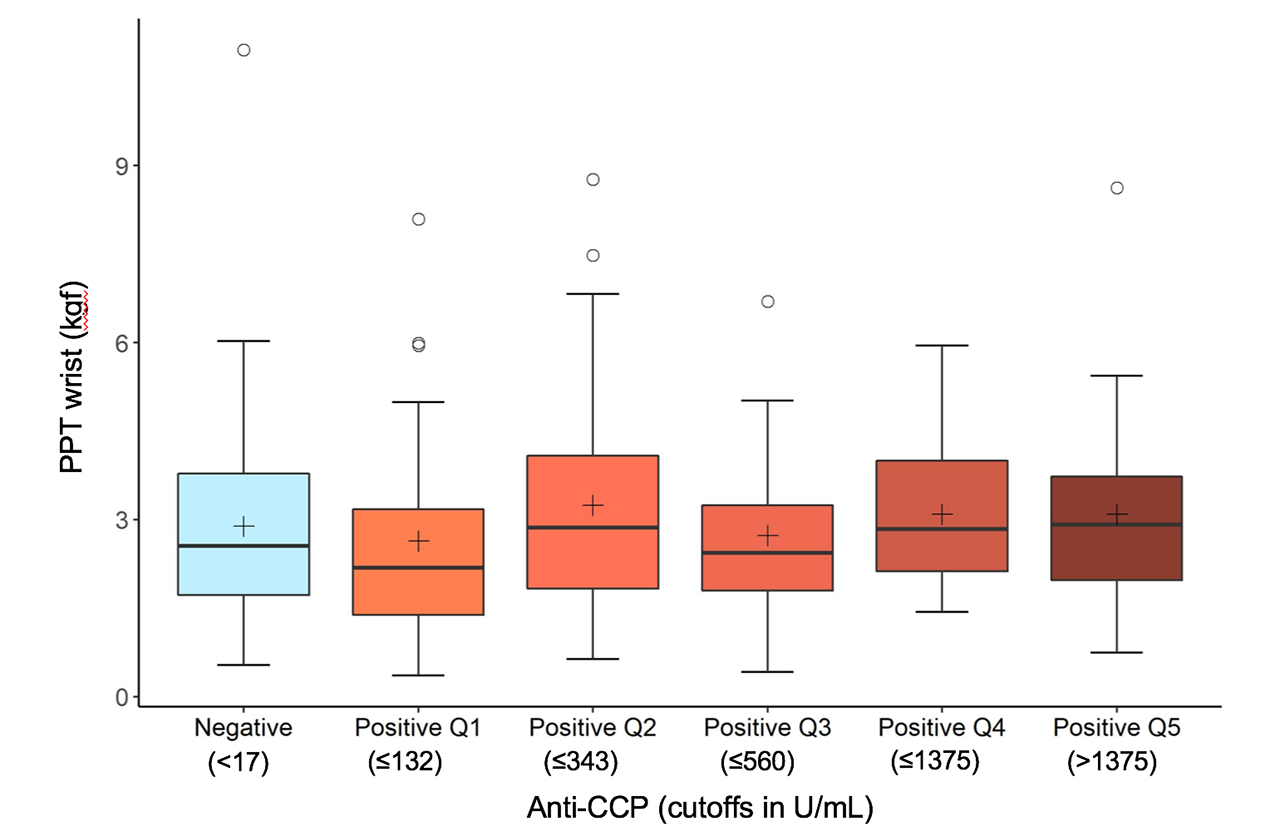
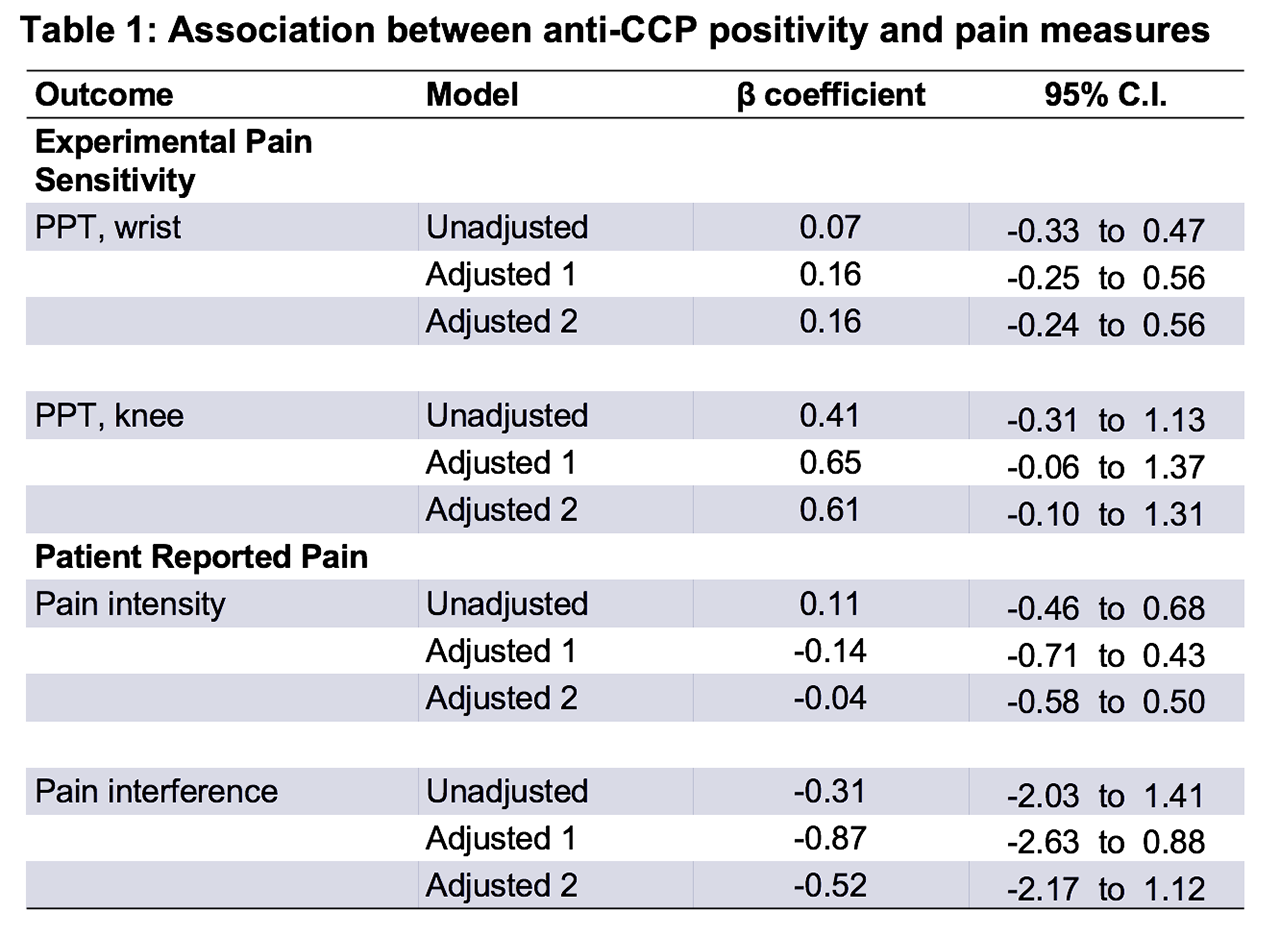
Results: There were 264 participants (mean age 54.6 years, average disease duration 10.2 years). On average, participants had 10.9 tender joints, 5.2 swollen joints, and a hsCRP level of 7.3 mg/L. Anti-CCP positivity was present in 164 patients (62.1%) and was not significantly associated with PPT at the wrist (b-coefficient 0.16, 95% confidence interval [CI] -0.24, 0.56) or knee (b-coefficient 0.61, 95% CI -0.10, 1.31) (Table 1, Figure 1). Similarly, there was no association between anti-CCP seropositivity and pain intensity (b-coefficient -0.04, 95% CI -0.58, 0.50) or pain interference (b-coefficient -0.52, 95% CI -2.17, 1.12). Categorizing seropositive anti-CCP into quintiles of increasing serum antibody did not show a dose-response relationship between anti-CCP and PPT at the wrist (Figure 2) or anti-CCP and any other pain measure (data not shown).
Conclusion: Among patients with active RA, anti-CCP seropositivity and higher levels of anti-CCP were not associated with experimental measures of pain sensitivity or patient-reported measures of pain. The possibility that untreated patients may demonstrate different findings cannot be ruled out. Nonetheless, these results contrast with data from animal models of inflammatory arthritis, as our data did not show evidence of an anti-CCP-antibody-mediated mechanism of joint pain as suggested in the animal models.
Adjusted 2: adjusted for covariates in Adjusted 1 + inflammation -hsCRP, swollen joint count- and fibromyalgia status.
To cite this abstract in AMA style:
Mun Y, Neogi T, Song J, Dunlop D, Heisler A, Bolster M, Bingham C, Marder W, Wohlfahrt A, Lee Y. Anti-CCP Antibody and Pain Sensitization in Rheumatoid Arthritis [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/anti-ccp-antibody-and-pain-sensitization-in-rheumatoid-arthritis/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/anti-ccp-antibody-and-pain-sensitization-in-rheumatoid-arthritis/