Session Information
Date: Monday, October 22, 2018
Title: Spondyloarthritis Including Psoriatic Arthritis – Clinical Poster II: Clinical/Epidemiology Studies
Session Type: ACR Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose: Obesity is associated with PsA risk, severity, and lack of response to DMARDs. However, no studies have explored adipose tissue inflammation at the molecular level and its associations with PsA features.
Methods: Periumbilical subcutaneous adipose tissue was obtained from 26 PsA patients without diabetes and compared with individuals with RA (n=42) and controls without rheumatic disease (n=21). Using real time polymerase chain reaction (RT-PCR), gene expression profiles for selected inflammatory cytokines, chemokines, macrophage and T cell markers, adipokines, and mediators of glucose and lipid metabolism were assessed and their correlations with PsA disease characteristics were compared between groups. Multivariable linear and logistic regression were used to adjust for relevant confounders.
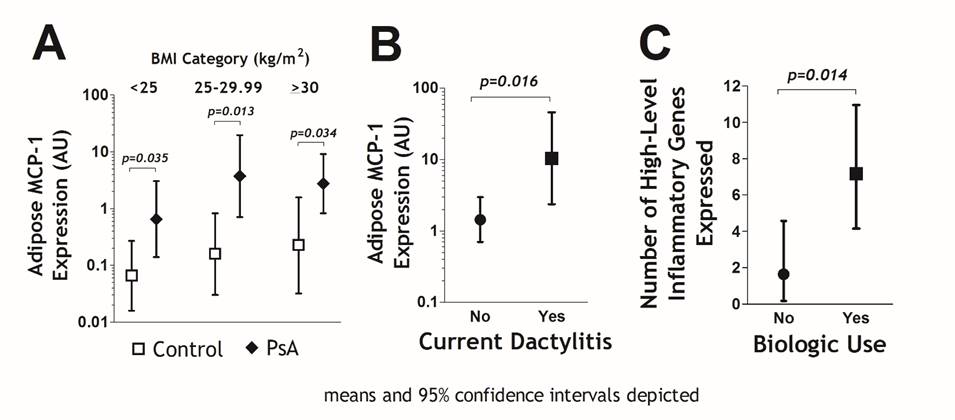
Results: The PsA group [54% female, mean age 56 years, mean BMI 31.1 kg/m2, median Composite Psoriatic Disease Activity Index(CPDAI)=4 units] did not differ significantly on age distribution compared with the RA or control groups. However, the PsA group was more frequently male and mean BMI was higher by 3.0-3.3 kg/m2. In general, adipose expression of inflammatory and metabolism genes was many fold higher in the PsA group compared with the RA and control groups, with 17 genes meeting the Bonferroni corrected threshold of p<0.002 [adiponectin, C3, MCP-1, CD4, HIF1a, IL-17R, IL-1, IL-4, IL-8, insulin receptor, leptin, NFkB, PPAR-gamma, PAI-1, osteopontin, STAT1, and TLR4], ranging from 4 to >450-fold higher in PsA vs. non-rheumatic controls, with little change after adjustment for sex and BMI. For each, higher expression was observed across all strata of BMI (Fig A depicted for MCP-1 as a representative example). Adipose MCP-1 expression was significantly associated with CPDAI (Spearman’s rho=0.402; p=0.042). CPDAI was also inversely associated with adipose GLUT4 expression, a key glucose transporter and indicator of insulin sensitivity (Spearman’s rho= – 0.434; p=0.030). The PsA feature with the strongest association with adipose MCP-1 expression was dactylitis (Fig B). Somewhat unexpectedly, those treated with biologics [n=18; TNF inhibitors (n=16), IL-17 inhibitors (n=2)] had more extensive adipose expression of inflammatory genes, even with adjusting for CPDAI, BMI, and demographics. Those using biologics had an average of 7.2 adipose inflammatory genes expressed at high level (i.e. >75th percentile) vs. an average of 1.6 genes among those not treated with biologics (p=0.014; Fig C).
Conclusion: PsA adipose tissue is a potent producer of inflammatory mediators that may contribute to disease phenotype. The potential for an off-target detrimental effect of biologics on adipose inflammation warrants additional study, particularly in light of reports of weight gain with TNF inhibitor use and a blunted effect of biologics among obese PsA patients.
To cite this abstract in AMA style:
Yun JH, Winchester R, Zhang HZ, Depender C, Giles JT. Adipose Tissue Inflammation in Psoriatic Arthritis: Overexpression of a Wide Array of Inflammatory Mediators and Associations with Disease and Treatment Characteristics [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 9). https://acrabstracts.org/abstract/adipose-tissue-inflammation-in-psoriatic-arthritis-overexpression-of-a-wide-array-of-inflammatory-mediators-and-associations-with-disease-and-treatment-characteristics/. Accessed .« Back to 2018 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/adipose-tissue-inflammation-in-psoriatic-arthritis-overexpression-of-a-wide-array-of-inflammatory-mediators-and-associations-with-disease-and-treatment-characteristics/