Session Information
Date: Monday, October 22, 2018
Title: Systemic Lupus Erythematosus – Clinical Poster II: Biomarkers and Outcomes
Session Type: ACR Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose:
Titers of HEp-2 ANAs vary between laboratories, with many contributing factors. We sought to systematically determine the contribution of different ANA kit manufacturers to ANA variation.
Methods:
Proficiency surveys involve sending aliquots of the same specimen to performing laboratories, allowing labs to compare results. Positive HEp-2 ANA titers reported by over 500 participant labs in ANA proficiency surveys by the College of American Pathologists between 2008 and 2017 were analyzed according to the kit manufacturer used by labs. The mean positive ANA titers reported for each specimen were ranked by kit manufacturer, relative to other manufacturers reporting on the same specimen. A total of 79 positive ANA specimens and up to 10 manufacturers were available for analysis, totaling 733 specimen-manufacturer combinations, with a median 46 laboratories reporting results for each combination.
Results: ANA titers of individual specimens differed by up to 5 2-fold dilutions (e.g. ANA 1:40 and 1:1280 reported by one or more laboratories testing the same specimen). The geometric mean titers for different methods differed from the overall mean titer by as much as 2-fold, and the difference between the geometric means of the highest method and the lowest method testing a given specimen over 3 2-fold dilution titers. ANA titer results were strongly influenced by the HEp-2 manufacture used by the labs (p<0.0001 by analysis of variance). Each manufacturer tended to have a fairly consistent rank, relative to the other manufacturers, (figure 1, median rank is indentation, diamond is mean for each manufacturer). Over the 10 years studied, the rank order of the ANA titer for each method was remarkably consistent (figure 2; each line tracks a manufacturer’s mean rank by year). The ANA patterns did not significantly affect these results.
Conclusion:
The variability in ANA titers is strongly influenced by the manufacturer of the HEp2 kit used in the assays and the differences between kits are generally consistent. Since much of the variability is systematic and associated with the kit manufacturer, harmonizing manufacturers’ kits could improve consistency of ANA reporting. 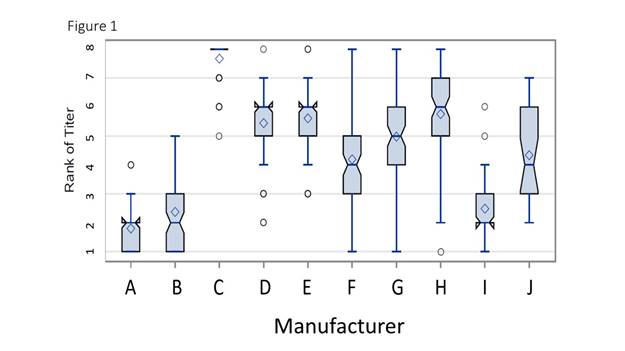

To cite this abstract in AMA style:
Wener M, Wener MH. Variation in HEp-2 Antinuclear Antibody (ANA) Titer Is Strongly Associated with the ANA Kit Manufacturer [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 9). https://acrabstracts.org/abstract/variation-in-hep-2-antinuclear-antibody-ana-titer-is-strongly-associated-with-the-ana-kit-manufacturer/. Accessed .« Back to 2018 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/variation-in-hep-2-antinuclear-antibody-ana-titer-is-strongly-associated-with-the-ana-kit-manufacturer/
