Session Information
Date: Monday, October 22, 2018
Title: Rheumatoid Arthritis – Diagnosis, Manifestations, and Outcomes Poster II: Diagnosis and Prognosis
Session Type: ACR Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose: Anti-cyclic citrullinated peptide (CCP) antibody test is a very important test for the diagnosis of rheumatoid arthritis (RA), but one of the disadvantages of the existing enzyme-linked immunosorbent assay (ELISA) is that its method is labor-intensive and time-consuming. The aim of this study was to assess the measurement characteristics of a rapid point-of-care (POC) anti-CCP test compared to the conventional anti-CCP test.
Methods: We used the POC ichroma anti-CCP test of Boditech Med and two anti-CCP assays (Elecsys¢ç Anti-CCP by Roche Diagnostics and the FCCP600 of Axis-Shield Diagnostics) and rheumatoid factor (RF) by latex agglutination test (LAT) respectively.
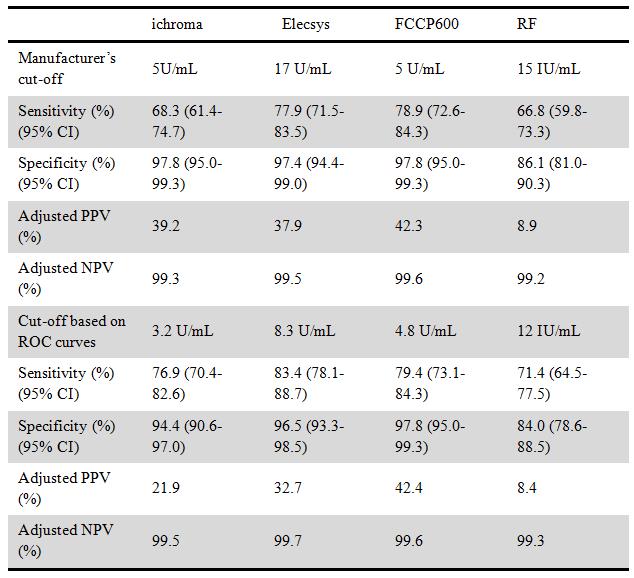
Results: A total of 430 subjects (199 RA patients, 112 non-RA patients and 119 healthy controls) were enrolled in this study. For the RA patients, the anti-CCP test were positive for 136 (68.3%), 155 (77.8%), and 158 patients (79.3%) with the ichroma anti-CCP, the Elecsys anti-CCP, and FCCP600, respectively. For the non-RA patients, the anti-CCP test were positive for 5 (4.5%), 7 (6.3%), and 5 patients (4.5%) with the ichroma anti-CCP, the Elecsys anti-CCP, and FCCP600, respectively. The overall agreement between the ichroma system and other tests was 91.6% (Elecsys) and 88.8% (FCCP) and 76.7% for RF. The difference between three areas under the ROC curves, including the POCT test, was not statistically significant (ichroma, 0.903 (0.874-0.933); Elecsys, 0.909 (0.877-0.941); and FCCP600, 0.892 (0.858-0.926)). There was, however, a significant difference from RF (0.770 (CI 0.721-0.819)).
Conclusion: The POC ichroma anti-CCP test is not only a quick and convenient test, but also provides a comparable and reliable diagnostic performance of conventional methods.
Table. Diagnostic performance of the three anti-CCP assays and one rheumatoid factor test for diagnosis of RA
Abbreviation : PPV, positive predictive value; NPV, negative predictive value
Fig. Scatter plots of anti-CCP level according to the concentrations of anti-CCP antibodies and rheumatoid factor for different groups of patients
To cite this abstract in AMA style:
Byun S, Mun C, Lee MS, Lee SW, Song JJ, Park YB. The Point-of-Care Ichroma Anti-CCP Test Showed a Competitive Measurement Accuracy, in Comparison to the Conventional Automated Tests [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 9). https://acrabstracts.org/abstract/the-point-of-care-ichroma-anti-ccp-test-showed-a-competitive-measurement-accuracy-in-comparison-to-the-conventional-automated-tests/. Accessed .« Back to 2018 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/the-point-of-care-ichroma-anti-ccp-test-showed-a-competitive-measurement-accuracy-in-comparison-to-the-conventional-automated-tests/