Session Information
Session Type: Poster Session (Monday)
Session Time: 9:00AM-11:00AM
Background/Purpose: The first symptoms of ankylosing spondylitis (AS) patients usually begin prior to 45 years, but can occur later in life. The purpose of this study is to evaluate the efficacy and safety of anti-TNFα treatment in late-onset AS (LoAS) patients in comparison to those with adult onset AS (AoAS).
Methods: We studied AS patients in TURKBIO registry between the dates of January 2011 and November 2018. All the patients fulfilled the modified New York criteria for AS and were classified into 2 groups based on their age at symptom onset: AoAS (age >16 but ≤45 years); and LoAS (age >45 years). In both groups, the following data were evaluated and compared: (1) epidemiological variables (2) clinical manifestations, including signs and symptoms at diagnosis; (3) laboratory results (4) disease activity markers and follow-up parameters (BASDAI, ASDAS-CRP and HAQ); (5) previous and current treatments (6) adverse events.
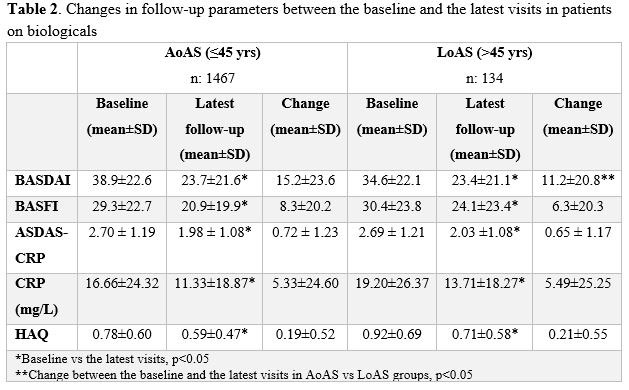
Results:
A total of 2551 AS patients (91,1% with AoAS and 8.9% with LoAS) were included in the study. LoAS group had more female patients, older age, shorter disease duration, shorter diagnostic delay, higher initial ESR and less HLA-B27 positivity compared to the AoAS (Table 1). Peripheral arthritis (not statistically significant) and dactylitis was seen more common in the LoAS. The frequency of other involvements was similar between the groups (Table1).The frequency of using drugs was similar between each groups although the use of glucocorticoids and sulphasalazine was more common in the LoAS. Switching from the first anti-TNFa treatment to the second one was more common in the AoAS. However, there was found no significant difference between the two groups in 2 or more switch ratios (Table 1). At the latest visit after the anti-TNFα therapy, the mean improvement in BASDAI was significantly higher in the AoAS (Table 2). A total of 10 (4.4%) serious adverse events were reported in LoAS and 39 (1.7%) in AoAS patients in the follow-up (HR: 2.62; 95% CI: 1.32–5.18). Severe infections were the most commonly seen serious adverse events (1.3% in LoAS and 0.8% in AoAS), followed by rash and allergic reactions (0.9% in LoAS and 0.3% in AoAS). Tuberculosis was observed in 2 patients (0.9%) in LoAS and 9 (0.4%) in AoAS, malignancy in 3 patients (1.3%) in LoAS and 6 (0.3%) in AoAS.
Conclusion: Our data showed that almost 8.9% of the patients with AS had late-onset of symptoms. The results suggested that LoAS patients might have different demographic, clinical features, disease activity parameters at baseline. The frequency of anti-TNFα use and response rate to the treatment was also similar in LoAS to those in AoAS patients. The patients with LoAS seem to have more common severe adverse events compared to the AOAS patients possibly related to their older age.
To cite this abstract in AMA style:
Uslu S, Can G, Cefle A, Yılmaz S, Kocaer S, Yüce İnel T, Gülle S, Koca S, Yolbaş S, Öztürk M, Senel S, Inanc N, Dalkılıç E, Gunduz O, Tufan A, Akar S, Birlik M, Sari I, Akkoç N, Onen F. The Efficacy and Safety of Anti-TNFα Treatment in Ankylosing Spondylitis Patients with Late Onset Compared to Those with Adult Onset; The Data from TURKBIO Registry [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/the-efficacy-and-safety-of-anti-tnf%ce%b1-treatment-in-ankylosing-spondylitis-patients-with-late-onset-compared-to-those-with-adult-onset-the-data-from-turkbio-registry/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/the-efficacy-and-safety-of-anti-tnf%ce%b1-treatment-in-ankylosing-spondylitis-patients-with-late-onset-compared-to-those-with-adult-onset-the-data-from-turkbio-registry/