Session Information
Date: Monday, October 22, 2018
Session Type: ACR Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose:
Interstitial lung disease (ILD) is a major cause of morbi-mortality in patients (pts) with CTD. Small studies have recently demonstrated a promising role for rituximab (RTX) in the treatment of CTD-ILD.
Methods:
We conducted a retrospective multicenter study including CTD-ILD pts treated with RTX. ILD was based on high resolution CT (HRCT) and/or lung biopsy. Results of HRCT, pulmonary function tests (PFTs) and 6-minute walking test (6MWT) before and after RTX were collected and compared using Wilcoxon matched pair test.
Results:
Forty-five pts were included (77.8% female, 62±12.9 years (yrs) old at last follow-up and mean CTD duration of 12.8±6.8 yrs). Eighteen pts (40%) were current/former smokers. Twenty-nine (64.4%) pts had RA, 4 (8.9%) primary SS, 3 (6.7%) SSc, 3 PM, 3 SLE, 1 (2.2%) DM, 1 antisynthetase syndrome and 1 overlap syndrome (SLE/SS). Among RA pts, 24 (82.8%) had positive RF and 28 (96.6%) ACPA. ANA were positive in 26/44 pts (57.8%).
ILD was diagnosed after 4yrs [IQR 1-9.5] of CTD. Only 1pt with overlap SLE/SS had ILD as a prior diagnosis. Non-specific interstitial pneumonia (NSIP) was present in 17 (37.8%) pts, usual interstitial pneumonia in 16 (35.6%), lymphocytic interstitial pneumonia in 2 (4.4%) and endogenous lipoid pneumonia in 1 (2.2%); 9pts had unspecific ILD pattern in HRCT.
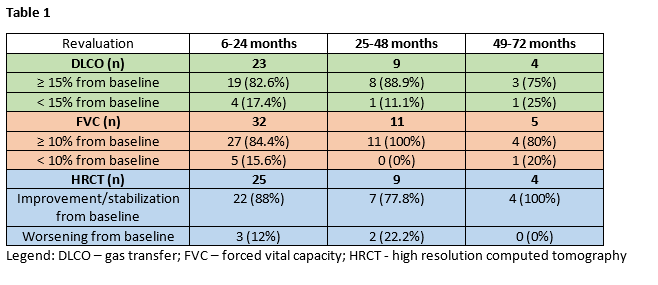
RTX was administered 1g twice, 2 weeks apart, with a median of 2 cycles [IQR 1-4]. Three pts received concomitantly AZA. Four pts were previously treated with CYC and/or MMF and/or AZA in association with steroids. The median interval between ILD diagnosis and first RTX administration was 1yr [IQR 0-4.5]. After 1yr on RTX there was a stabilization in gas transfer (+7.1%, p=0.15) and in forced vital capacity (+3%, p=0.49) in the whole group. At last follow-up (median 3yrs [IQR 1-6] after starting RTX), 30/31 pts (36.8%) had stabilized/improved dyspnea according to New York Heart Association criteria. Data on 6MWT were lacking for proper conclusions. Detailed responses to RTX are shown in table 1.
Thirteen (28.9%) pts stopped RTX, with infection being the main cause (4pts; none had hypogammaglobulinemia; 2 receiving concomitant LFN and 1 AZA). Infusion reaction, uncontrolled joint disease, suspected lung cancer and long-standing stable disease led to RTX suspension in 2pts each. Five pts died, 2yrs (IQR 1-5.25] after CTD diagnosis. One pt concomitantly treated with MTX died 1week after the first RTX cycle due to chest infection; in the other pts the cause was unknown.
Conclusion:
Our results reinforce the promising role of RTX in a wide range of CTD-ILD pts and demonstrate an association with long-standing disease stability, particularly in pts with NSIP (7/11 pts with FVC≥10% from baseline after more than 24 months on RTX had NSIP). Four pts suspended RTX and 1 died due to infection, making monitoring and prophylaxis (vaccines) of extreme importance, particularly in pts with underlying ILD.
To cite this abstract in AMA style:
Duarte AC, Cordeiro A, Fernandes B, Bernardes M, Tenazinha C, Cordeiro I, Santiago T, Seixas MI, Roxo Ribeiro A, Santos MJ. Rituximab in Connective Tissue Disease – Associated Interstitial Lung Disease [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 9). https://acrabstracts.org/abstract/rituximab-in-connective-tissue-disease-associated-interstitial-lung-disease/. Accessed .« Back to 2018 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/rituximab-in-connective-tissue-disease-associated-interstitial-lung-disease/