Session Information
Session Type: ACR Abstract Session
Session Time: 4:30PM-6:00PM
Background/Purpose:
Acute anterior uveitis (AAU), inflammation of the anterior uveal tract, is the most common extraarticular manifestation in patients (pts) with axial spondyloarthritis (axSpA), reported by up to 40% of pts.1 Uveitis is associated with a significant clinical burden; common symptoms include blurred vision, photophobia and pain.2 Previous studies have shown that anti-TNFs can reduce the incidence of AAU flares in pts with radiographic axSpA (ankylosing spondylitis),3,4,5 but few have focused on pts across the full axSpA spectrum. The aim of the C-VIEW study was to analyze the impact of certolizumab pegol (CZP) treatment on AAU flares in pts with active axSpA (radiographic and non-radiographic) and a recent history of AAU.
Methods: C-VIEW (NCT03020992) is an ongoing multicenter, open-label, phase 4 study. Pts had active axSpA according to the Assessment of SpondyloArthritis international Society (ASAS) classification criteria, a history of recurrent AAU (≥2 AAU flares in total and ≥1 AAU flare in the year prior to study entry), were HLA-B27 positive, and were eligible for anti-TNF treatment (active axSpA, previous failure of ≥2 NSAIDs, biologic naïve or had failed at most one anti-TNF). Pts received CZP 400 mg at Weeks (Wks) 0/2/4, then 200 mg every 2 wks through 96 wks. The primary variable was the incidence of AAU flares compared to historic rates. A pre-specified interim analysis compared AAU incidence in the 48 wks prior to CZP treatment initiation with the 48 wks of treatment, using Poisson regression adjusted for possible within-patient correlations, with period (pre- and post-baseline) and axSpA disease duration as covariates. Incidence rates (IR) were calculated based on the number of cases per pts at risk over 48 weeks. Observed data are reported.
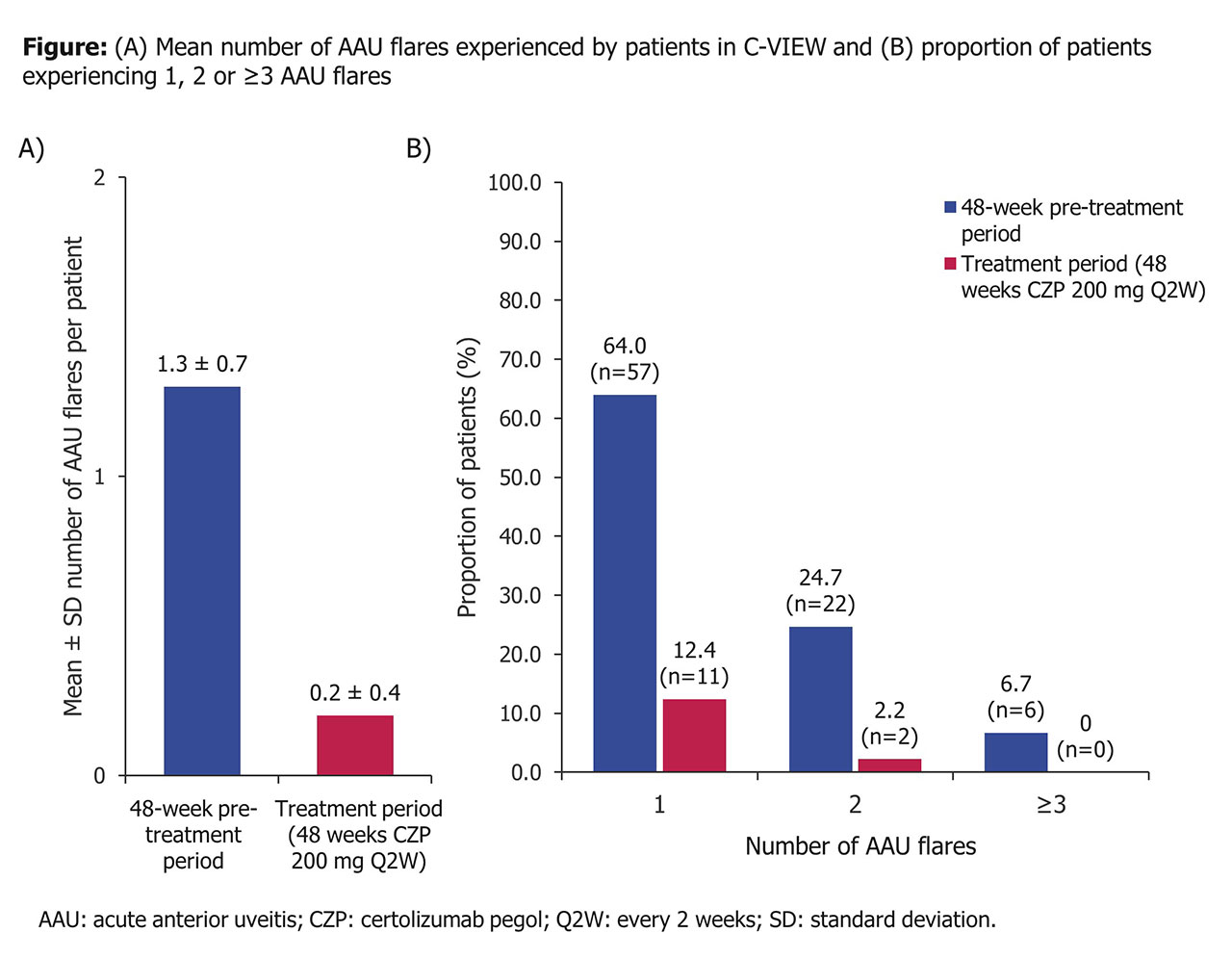
Results: Of 115 enrolled pts, 89 initiated CZP treatment and 85 completed Wk 48. Baseline characteristics are shown in the Table. The 48-wk interim analysis revealed significantly fewer AAU flares per pt during CZP treatment compared to before treatment (Figure; Poisson-adjusted IR: 0.2 vs 1.5, p< 0.001). The number of pts experiencing 1 and ≥2 AAU flares (64.0% and 31.5%, respectively) was substantially reduced during CZP treatment (12.4% and 2.2%). In the 13 patients who had AAU flares both pre- and post-baseline, the mean duration of AAU flares was also reduced during CZP treatment from 97.4 to 58.4 days. After 48 wks CZP, pts’ disease activity had improved substantially (mean ± SD Ankylosing Spondylitis Disease Activity Score [ASDAS]: 2.0 ± 0.9; BASDAI: 3.3 ± 2.1), with 31.4% of pts achieving ASAS partial remission and 29.1% ASDAS major improvement. No new safety signals were identified.
Conclusion:
In this open-label study, we found a significant reduction in the AAU flare rate in axSpA pts with a history of recurrent AAU during the first 48 wks of CZP treatment. Pts also experienced significant improvement in axSpA disease activity during CZP treatment.
References:
1. Martin TM. Curr Opin Rheumatol 2002;14:337–41; 2. Bacchiega ABS. Rheumatology (Oxford) 2017;56:2060–7; 3. van der Heijde D. Rheumatology (Oxford) 2017;56:1498–1509; 4. van Bentum RE. J Rheumatol 2019;46:153–9; 5. van Denderen JC. J Rheumatol 2014;41:1843–8.
To cite this abstract in AMA style:
van der Horst-Bruinsma I, van Bentum R, Verbraak F, Rath T, Rosenbaum J, Misterska-Skóra M, Hoepken B, Irvin-Sellers O, VanLunen B, Bauer L, Rudwaleit M. Reduction of Anterior Uveitis Flares in Patients with Axial Spondyloarthritis Following 1 Year of Treatment with Certolizumab Pegol: 48-Week Interim Results from a 96-Week Open-Label Study [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/reduction-of-anterior-uveitis-flares-in-patients-with-axial-spondyloarthritis-following-1-year-of-treatment-with-certolizumab-pegol-48-week-interim-results-from-a-96-week-open-label-study/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/reduction-of-anterior-uveitis-flares-in-patients-with-axial-spondyloarthritis-following-1-year-of-treatment-with-certolizumab-pegol-48-week-interim-results-from-a-96-week-open-label-study/