Session Information
Session Type: ACR Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose: Apremilast (APR), an oral phosphodiesterase 4 inhibitor, acts to regulate immune activity in psoriatic arthritis (PsA) patients. PALACE 1 (NCT01172938), 2 (NCT01212757), and 3 (NCT01212770) compared the efficacy and safety of APR with placebo (PBO) in patients with active PsA despite prior conventional disease-modifying anti-rheumatic drugs (DMARDs) and/or biologics. This pooled analysis of PALACE 1-3 assessed the long-term safety of APR treatment in patients treated for up to 3 years.
Methods: Patients were randomized (1:1:1) to receive PBO, APR 30 mg BID (APR30), or APR 20 mg BID (APR20) stratified by baseline DMARD use (yes/no). The PBO-controlled phase continued to Week 24; PBO patients were re-randomized to APR30 or APR20 at Week 16 (early escape) or Week 24. Double-blind APR treatment continued to Week 52; patients could continue APR during an open-label, long-term treatment phase. Visits in years 2 and 3 were scheduled at 13-week intervals.
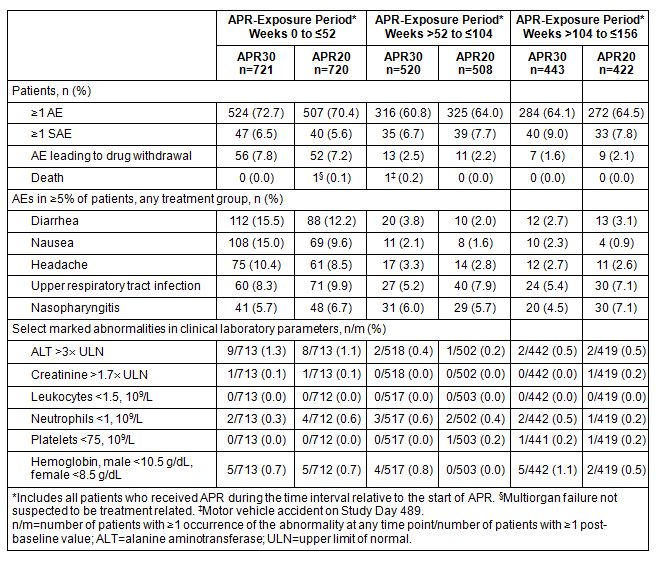
Results: A total of 1,493 patients were randomized and received ≥1 dose of study medication (PBO: n=495; APR30: n=497; APR20: n=501). At the 3-year data cut for PALACE 1-3, 1,441 patients in Weeks 0 to ≤52, 1,028 in Weeks >52 to ≤104, and 865 in Weeks >104 to ≤156 received APR. During the Weeks 0 to ≤52 APR-exposure period, adverse events (AEs) occurring in ≥5% of APR-exposed patients were diarrhea, nausea, headache, upper respiratory tract infection, and nasopharyngitis (Table). Most diarrhea and nausea events were reported within the first 2 weeks of treatment and usually resolved in 4 weeks without medical intervention. The frequency of gastrointestinal AEs decreased with longer exposure (i.e., Weeks >52 to ≤104 or >104 to ≤156; Table); other AEs occurring at ≥5% in the initial period either decreased in frequency or remained stable with prolonged exposure (Table). Most AEs were mild or moderate in severity for up to 156 weeks of APR exposure. Rates of depression remained very low, consistent with earlier findings. The rate of serious AEs (SAEs) was 8.4% during Weeks >104 to ≤156 of APR exposure; most SAEs occurred in 1 patient each. Rates for SAEs of special interest (major cardiac events, malignant neoplasms, serious opportunistic infections) were very low and comparable to those in the first year of treatment. The discontinuation rate due to AEs was 1.8% during Weeks >104 to ≤156 of APR exposure. Marked laboratory abnormalities were infrequent, and most returned to baseline with continued treatment.
Conclusion: APR demonstrated a favorable safety profile and was well tolerated for up to 156 weeks. The incidence of AEs remained stable or decreased with long-term exposure to APR.
Acknowledgment: We thank Adewale O. Adebajo for his work on the original abstract.
To cite this abstract in AMA style:
Mease PJ, Gladman DD, Gomez-Reino JJ, Hall S, Kavanaugh A, Lespessailles E, Schett G, Shah K, Teng L, Wollenhaupt J. Long-Term (156-Week) Safety Profile of Apremilast, an Oral Phosphodiesterase 4 Inhibitor, in Patients with Psoriatic Arthritis: Pooled Safety Analysis of 3 Phase III, Randomized, Controlled Trials [abstract]. Arthritis Rheumatol. 2016; 68 (suppl 10). https://acrabstracts.org/abstract/long-term-156-week-safety-profile-of-apremilast-an-oral-phosphodiesterase-4-inhibitor-in-patients-with-psoriatic-arthritis-pooled-safety-analysis-of-3-phase-iii-randomized-controlled-trials/. Accessed .« Back to 2016 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/long-term-156-week-safety-profile-of-apremilast-an-oral-phosphodiesterase-4-inhibitor-in-patients-with-psoriatic-arthritis-pooled-safety-analysis-of-3-phase-iii-randomized-controlled-trials/