Session Information
Session Type: Abstract Submissions (ACR)
Background/Purpose: Immunoglobulin G receptors (FcγR) are critical in the development of autoimmunity and pathogenesis of immune complex (IC) diseases. Although ICs activate complement, contributing to tissue damage, it is reported complement does not affect IC interaction with FcγR. We have re-explored this issue and find complement has a major impact on IC interaction with FcγR. Specifically, the classical pathway (CP) down-regulates and the alternative pathway (AP) promotes IC binding to FcγR. We performed reverse passive Arthus reactions, a model of IC-mediated cutaneous vasculitis, in normal C57Bl/6 mice and mice genetically deficient in complement proteins C1q, C4 and Factor B. It is also reported that complement does not contribute to Arthus reactions in mice, unlike in other animals. We hypothesized the CP, rather than contributing to vasculitis, down-regulates tissue injury caused by IC interaction with activating FcγR; therefore, its absence (C1q-/- and C4-/-) would enhance cutaneous vasculitis, while absence of the AP (Factor B-/-) would diminish cutaneous vasculitis.
Methods: Sedated and shaved mice (normal, C1q-/-, C4-/-, and Factor B-/-) were injected intradermally with 20 µl of PBS alone on one side and affinity purified rabbit anti-bovine serum albumin (BSA) IgG 5 µg in PBS on the opposite side. Immediately following, BSA 100 µg and 125Iodine-labeled BSA 1.25 µg in PBS containing 1% Evans blue was injected intravenously. After 4 hours, mice were sacrificed and the extravasated blue spot and control injection spot were dissected and weighed. Radioactivity per specimen weight (cpm/gm) of treated skin was divided by that of control skin to determine an Arthus index (AI) for each mouse. Arthus indices were first analyzed by Kruskal-Wallis ranked test, followed by Mann-Whitney U test to evaluate for significant differences in median AI. P values less than 0.05 were considered significant.
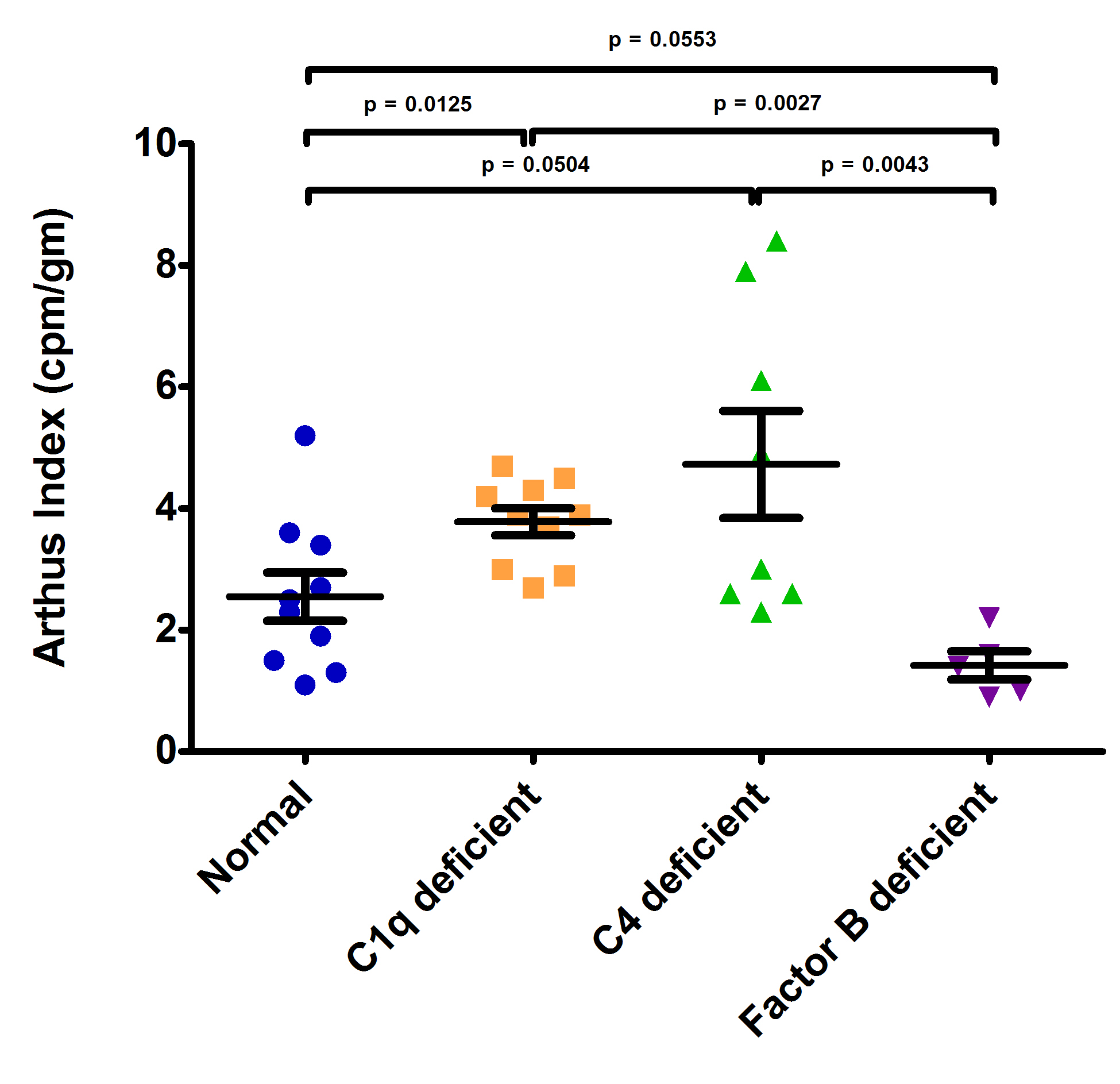
Results: Complement activation strongly influenced the degree of cutaneous vasculitis in mice (Fig. 1, p=0.0013). C1q-/- (mean AI 3.8±0.7, p=0.0125) and C4-/- (mean AI 4.7±2.3, p=0.0504) mice exhibited more extensive vasculitis than normal animals (mean AI 2.6±1.2). Conversely, Factor B deficient (mean AI 1.4±0.5, p=0.0553) mice trended towards significantly reduced vasculitis compared to normal animals.
Conclusion: Cutaneous vasculitis is significantly greater in mice deficient in CP complement proteins and appeared reduced in mice deficient in AP complement proteins. Our results are the first to provide physiologic evidence in vivo that complement critically influences IC and FcγR mediated inflammation. They also refute previous data suggesting complement possesses no role in Arthus-induced vasculitis in mice. They may further our understanding of why individuals with CP defects are noted to have frequent signs of autoimmune pathology.
Fig 1: Reverse Passive Arthus Reactions
Disclosure:
E. Y. Wu,
None;
H. Jiang,
None;
C. G. Hester,
None;
M. M. Frank,
None.
« Back to 2012 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/immunoglobulin-g-fc-receptor-activity-in-vivo-is-under-complement-control/