Session Information
Date: Monday, November 9, 2015
Title: Systemic Lupus Erythematosus - Clinical Aspects and Treatment Poster Session II
Session Type: ACR Poster Session B
Session Time: 9:00AM-11:00AM
Background/Purpose: Hypogammaglobulinemia has been
associated with serious infectious adverse events (SIAE) and may occur during
immunosuppressive therapy for lupus nephritis (LN). It is possible that proteinuria
contributes to low Ig levels, but this relationship has not been explored. We analyzed
data from the Abatacept and Cyclophosphamide Combination Therapy for Lupus
Nephritis (ACCESS) trial to evaluate the relationship between proteinuria and IgG
levels in patients undergoing treatment for LN with the Euro-Lupus Nephritis (ELN)
regimen.
Methods: The ACCESS trial compared abatacept to
placebo in LN patients undergoing ELN treatment with cyclophosphamide followed
by azathioprine. Shortly after the trial began, the protocol was amended to
include quantitative Ig measurements at baseline and weeks 2, 4, 12, and 24 in
all subsequent participants (n=102). For the purpose of this analysis, hypogammaglobulinemia
was defined by an IgG level <450 mg/dL based on previous studies indicating
that this level of IgG is associated with an increased infection risk in people
with hereditary immunodeficiency. Urine protein-to-creatinine ratio (UPCR) was
calculated using 24-hour urine collections. SIAE were included if they were
grade 3 or higher or were associated with hospitalization. Fisher’s exact test
was used to evaluate categorical relationships, and correlation and linear
regression were used to assess continuous relationships between measurements.
Results: Linear regression analysis showed an
inverse correlation between proteinuria and IgG levels (r=-0.42, p<0.0001). At
baseline, 24% of participants with UPCR ≥3
had IgG<450 mg/dL, compared with 5% of participants with UPCR <3 (p=0.013).
Eleven participants experienced SIAE during treatment. None of the SIAE
occurred among the 13 participants with low baseline IgG, compared to 11/89 participants
with IgG ≥450 at baseline (0% vs. 12%).
Overall, 31 participants had transiently low IgG (<450 mg/dL) at some time
during treatment, whereas 71 participants never experienced low IgG. SIAE occurred
in 3/31 (10%) participants who had low IgG at some point vs. 8/71 (11%) participants
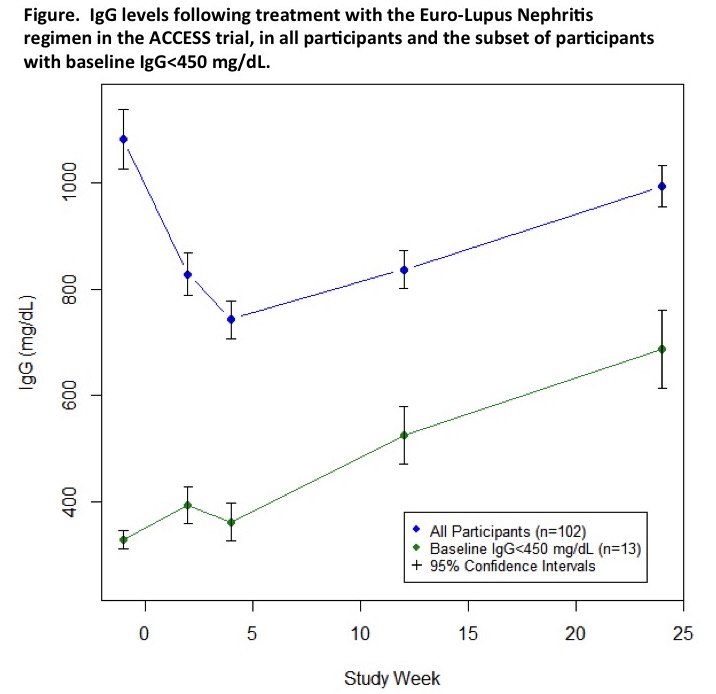
who never had low IgG. Mean IgG levels were lowest approximately 4 weeks
following initiation of therapy and then rose (see Figure), so that only 1/78 participants
who reached the primary study endpoint at week 24 had IgG <450 mg/dL at that
point. Results in the abatacept and placebo subsets were similar.
Conclusion: Our results demonstrate an inverse
relationship between proteinuria and low IgG levels in LN. However, low IgG was
not associated with an increased risk of SIAE in the ACCESS trial, even among participants
with IgG <450 mg/dL. Moreover, among participants who completed 24 weeks of
ELN treatment, hypogammaglobulinemia corrected in all but one subject. These
findings suggest that low IgG levels should not be a contraindication to immunosuppressive
treatment of active LN.
To cite this abstract in AMA style:
Murray SG, Lim N, Stahly M, Smilek D, Wofsy D. IgG Levels Correlate Inversely with Proteinuria Among Participants in the Abatacept and Cyclophosphamide Combination Therapy for Lupus Nephritis Trial, but Hypogammaglobulinemia Was Not Associated with an Increased Risk of Serious Infection [abstract]. Arthritis Rheumatol. 2015; 67 (suppl 10). https://acrabstracts.org/abstract/igg-levels-correlate-inversely-with-proteinuria-among-participants-in-the-abatacept-and-cyclophosphamide-combination-therapy-for-lupus-nephritis-trial-but-hypogammaglobulinemia-was-not-associated-wit/. Accessed .« Back to 2015 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/igg-levels-correlate-inversely-with-proteinuria-among-participants-in-the-abatacept-and-cyclophosphamide-combination-therapy-for-lupus-nephritis-trial-but-hypogammaglobulinemia-was-not-associated-wit/