Session Information
Session Type: Abstract Submissions
Session Time: 5:30PM-7:00PM
Background/Purpose: The efficacy and safety of intravenous (IV) tocilizumab (TCZ), an interleukin-6 receptor-alpha inhibitor, have been demonstrated in patients (pts) with polyarticular-course juvenile idiopathic arthritis (pcJIA) (Brunner HI et al. Ann Rheum Dis. 2014;74:1110-7). This study investigated appropriate dosing regimens of subcutaneous (SC) TCZ in pts with pcJIA.
Methods: We enrolled pts aged 1-17 years with pcJIA and previous inadequate response/intolerance to methotrexate who were TCZ naive or were receiving TCZ IV with adequate disease control. TCZ SC was administered open label according to a body weight (BW)–based dosing regimen designed following PK simulations of the approved IV dosing in pcJIA and SC dosing in adult rheumatoid arthritis: pcJIA pts weighing <30 kg received TCZ 162 mg every 3 weeks (Q3W) and pcJIA pts weighing ≥30 kg received TCZ 162 mg Q2W for 52 weeks. Model-computed pharmacokinetic (PK) and pharmacodynamic (PD) parameters and safety and efficacy (exploratory) were assessed.
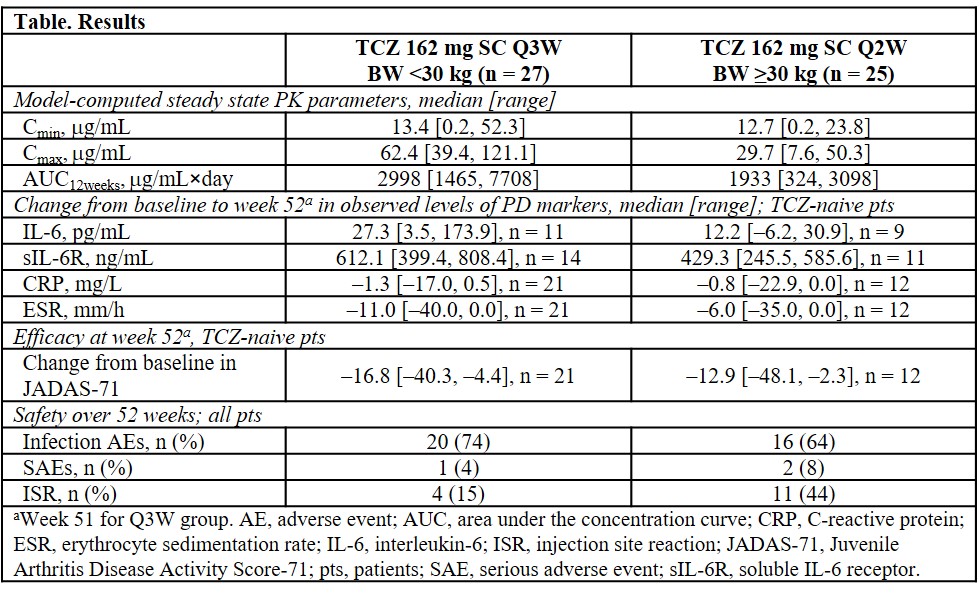
Results: Fifty-two pts were enrolled; 27 <30 kg BW received TCZ 162 mg SC Q3W and 25 ≥30 kg BW received TCZ 162 mg SC Q2W. Overall, 69% of pts were female. Median baseline age was 6.0 years for the <30 kg BW group and 15.0 years for the ≥30 kg BW group; 85% and 56%, respectively, were TCZ naive. Because no notable differences in steady state PK occurred between naive vs non-naive pts, pooled data are presented. Median Cmin was similar between BW groups and higher than previously established with TCZ IV (TCZ IV median Cmin: 3.2 μg/mL for TCZ 10 mg/kg <30 kg BW and 7.3 μg/mL for TCZ 8 mg/kg ≥30 kg BW), ensuring adequate exposure from SC doses. Median Cmax from SC dosing was lower than from IV dosing in both BW groups (TCZ IV median Cmax: 191 μg/mL for TCZ 10 mg/kg <30 kg BW and 201 μg/mL for TCZ 8 mg/kg ≥30 kg BW). Cmax and AUC12weeks were higher with TCZ SC in the <30 kg BW group than the ≥30 kg BW. Changes in PD parameters for TCZ-naive pts were consistent with those previously observed for TCZ IV and suggest improvement of inflammation. JADAS-71 generally improved in both BW groups (Table), with trends consistent with those observed for TCZ IV. Infections were the most frequent adverse event (AE), reported in 20 pts in the <30 kg BW group and 16 pts in the ≥30 kg BW group (Table). Injection site reactions were more common in the ≥30 kg BW group (Table). The most common symptoms were erythema, swelling, hematoma, pain, and pruritus. No serious hypersensitivity, AE leading to withdrawal, opportunistic infection, serious hepatic AE, or death occurred. There were 2 serious AEs (croup and varicella) in 1 pt in the <30 kg group and 2 serious AEs (anorexia and arthralgia) in 2 pts in the ≥30 kg group; the overall rate was 7.9/100 pt-years, consistent with that for TCZ IV.
Conclusion: The BW-based TCZ SC dosing regimens for pcJIA provided adequate exposure to support efficacy comparable to that of TCZ IV, with an acceptable benefit-risk profile.
To cite this abstract in AMA style:
Brunner H, Ruperto N, Martini A, Ramanan A, Cuttica R, Weiss JE, Henrickson M, Schmeling H, Anton J, Minden K, Hsu J, Bharucha K, Wimalasundera S, Kadva AK, Upmanyu R, Mallalieu NL, Lovell D, De Benedetti F. Identification of Optimal Subcutaneous Doses of Tocilizumab in Children With Polyarticular-Course Juvenile Idiopathic Arthritis [abstract]. Arthritis Rheumatol. 2017; 69 (suppl 4). https://acrabstracts.org/abstract/identification-of-optimal-subcutaneous-doses-of-tocilizumab-in-children-with-polyarticular-course-juvenile-idiopathic-arthritis/. Accessed .« Back to 2017 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/identification-of-optimal-subcutaneous-doses-of-tocilizumab-in-children-with-polyarticular-course-juvenile-idiopathic-arthritis/