Session Information
Session Type: ACR Poster Session A
Session Time: 9:00AM-11:00AM
Background/Purpose: The giant cell arteritis (GCA) management requires the administration of high-dose glucocorticoid (GC) at the introduction remission and the gradual reduction of the dosage. However, the long-term usage of GC can cause several side effects such as diabetes, osteoporosis and infections. Since patients with GCA are mainly elderly, the risk of side effects due to high doses or long-term administration of GC is greater. However, in clinical practice, the protocol of tapering GC depends on the judgment of an individual clinician. There is no research describing the clinical practice of GCA and GC tapering. Therefore, this study describes the background of patients with GCA and its management using the nationwide clinical database in Japan.
Methods: This was a retrospective cohort study using the Diagnosis Procedure Combination (DPC) database, a nationwide inpatient database of Japan. We identified patients who received treatment for GCA (ICD10 code: M316) between 2010 and 2013 and tracked patient data up to 180 days after hospitalization and confirmed the treatment situation of GCA.
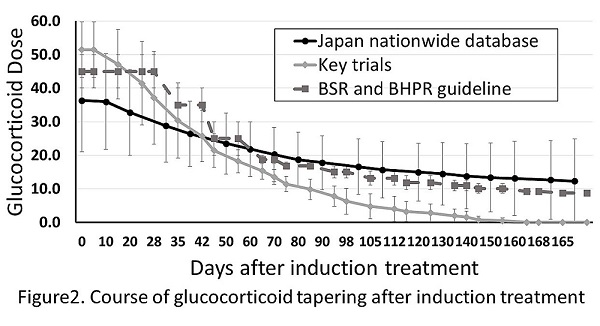
Results: 875 patients were hospitalized for GCA treatment from 2010 to 2013., 135 patients with age less than 50 years or no GC administration were excluded from the study and 740 patients were analyzed. 354 patients were followed until 90 days after admission and 220 patients were followed until 180 days after admission (Figure 1). The average patient age was 75 years, the mean body weight was 52 kg, and the complications were 27.6% (204/740) of diabetes, 10.4% (77/740) of gastrointestinal ulcers, and 6.8% (50/740) of malignancy. The GC dosage at the start of the treatment was 36.4 mg of prednisolone. The usage of pulse GC and immunosuppressant was 14.2% (105/740) and 11.6% (86/740), respectively. Of all, 69.7% (516/740) were prevented from osteoporosis. Although the mean GC dosage at the beginning was less than the key study protocol average and the recommended guideline, the dosage of GC after 90 days of admission was high (17.6 vs. 7.8 vs. 15 mg; Figure 2). The patientsf age (>74 years) significantly affected the GC dosage reduction after 90 days of admission (P < 0.045)). However, none of the hospital type (university hospital or an educational institution approved by the Japan College of Rheumatology), immunosuppressant combination, patient comorbidity affected the GC tapering at 90 days.
Conclusion: Many patients with GCA present with comorbidities. However, the tapering of GC dosage was slower than the main study protocol. Prophylaxis of complications due to GCs was not sufficiently undertaken.
To cite this abstract in AMA style:
Uechi E, Fushimi K. Epidemiological Study of Giant Cell Arteritis Using a Japanese Administrative Database [abstract]. Arthritis Rheumatol. 2017; 69 (suppl 10). https://acrabstracts.org/abstract/epidemiological-study-of-giant-cell-arteritis-using-a-japanese-administrative-database/. Accessed .« Back to 2017 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/epidemiological-study-of-giant-cell-arteritis-using-a-japanese-administrative-database/