Session Information
Date: Sunday, November 10, 2019
Title: Patient Outcomes, Preferences, & Attitudes Poster I: Patient Reported Outcomes
Session Type: Poster Session (Sunday)
Session Time: 9:00AM-11:00AM
Background/Purpose: RAPID3 is a disease activity measure which relies on patient-reported outcomes (PROs) to assess physical function, pain, and global health but does not require joint count or lab values. Patient Reported Outcomes Measurement Information System (PROMIS) is a validated instrument used to collect PROs across multiple diseases. Compared to RAPID3, PROMIS offers the ability to assess more targeted domains such as fatigue, global mental health, and pain interference. It also allows comparison of patient responses to a normalized US population. The purpose of this study was to assess whether changes in PROMIS domains correlate with changes in RAPID3 in RA patients starting a new biologic DMARD and whether PROMIS could complement a composite measure such as RAPID3 to optimize patient outcomes.
Methods: A retrospective chart review was conducted of RA patients in a tertiary outpatient rheumatology practice starting a new biologic DMARD. Computer Adaptive Testing (CAT) PROMIS domains (global physical health, global mental health, physical function, pain interference, and fatigue) were collected along with RAPID3 at baseline and follow-up visits. CAT uses item response theory and branching logic to quickly assess patient scores. Correlations between PROMIS domains and RAPID3 were calculated using Spearman’s correlations.
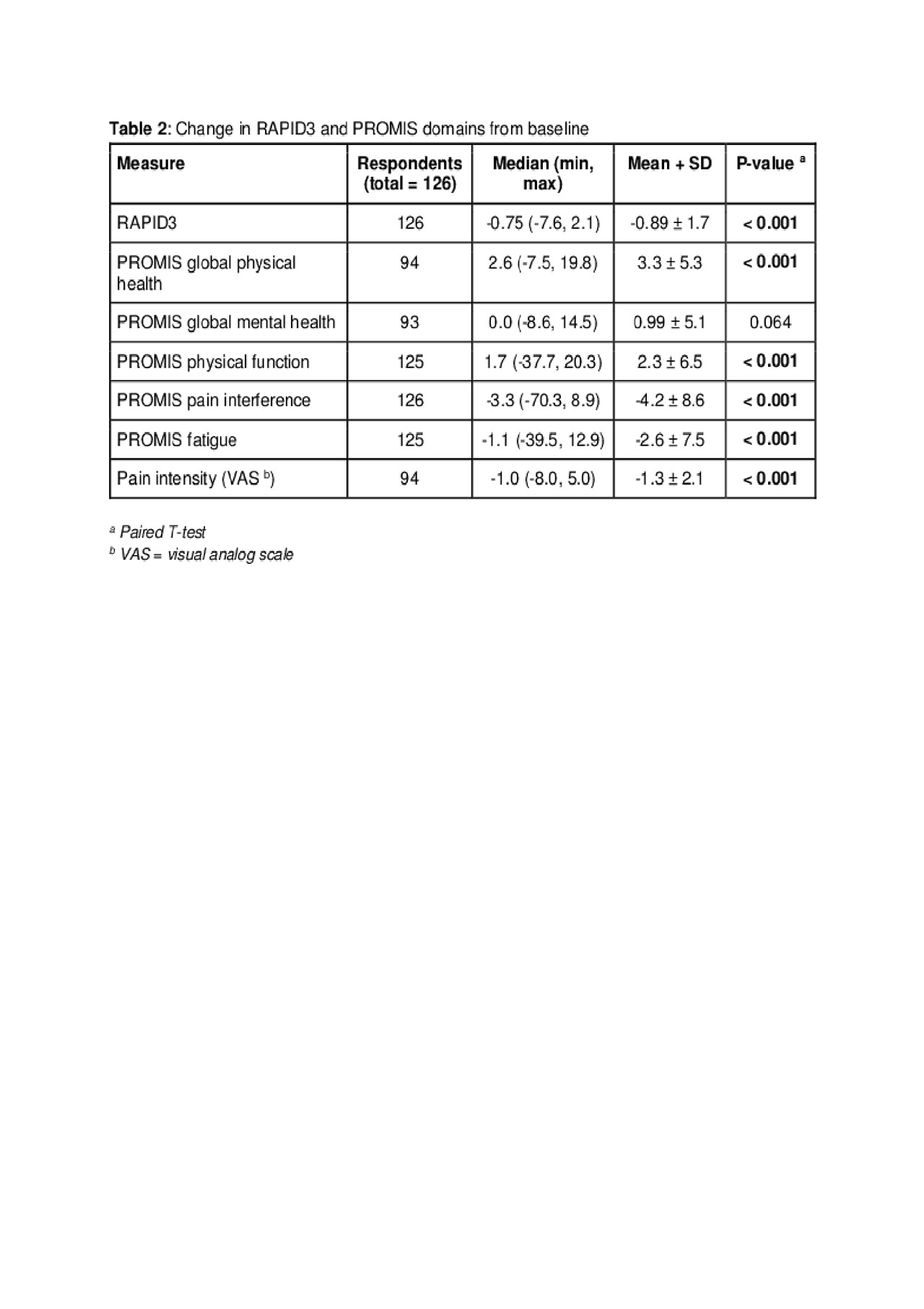
Results: 126 patients were included in the final analysis. Mean age was 56.2 ± 13.8 years and 75% were female. Disease duration was greater than 2 years in 80% of patients. 81% of patients had been on at least 1 or 2 prior DMARDs. All PROMIS domains, with the exception of global mental health, were significantly associated with changes in RAPID3 (p ≤ 0.05) and the coefficients suggest moderate correlation (|r| = 0.48 – 0.60). Number of days between initial and follow-up visit was 111.7 ± 48.6. There was no significant difference found between a follow-up period < 90 days compared to a follow-up period > 90 days. At follow up, patients in near remission by RAPID3 (≤ 1.3, n=12) had a PROMIS physical global health median score of 52.5 (min, max = 37.4, 61.9), while patients with high severity RAPID3 (≥ 4.3, n=55) had a PROMIS global health median of 34.9 (min, max = 26.7, 47.79; p< 0.001). A change of > 1.2 for RAPID3 and > 5 for PROMIS domains has been considered the minimal clinically important difference. 52% of patients had a RAPID3 change of >1.2. Of these, 80% had a change of > 5 in PROMIS pain interference, while 76%, 65%, and 59% had changes of > 5 in PROMIS fatigue, physical function, and global physical health respectively.
Conclusion: In RA patients starting a new biologic DMARD, changes in all studied PROMIS domains, with the exception of global mental health, were significantly associated with changes in RAPID3. Compared to RAPID3, use of PROMIS can allow for the assessment of additional domains in a format which is normalized to a broad population. Insight gained from these additional domains can help focus follow-up visits and facilitate joint decision making between patients and providers. A composite index of PROMIS domains predictive of changes in RAPID3 is being developed. Additional studies are needed to validate the use of PROMIS as a tool for disease activity measure in RA patients.
To cite this abstract in AMA style:
Williams T, Husni M, Jin Y, Strnad G, O'Bryant S, Deal C. Correlation Between Patient Reported Outcomes Measurement Information System (PROMIS) and RAPID3 in Rheumatoid Arthritis Patients Starting a New Biologic DMARD [abstract]. Arthritis Rheumatol. 2019; 71 (suppl 10). https://acrabstracts.org/abstract/correlation-between-patient-reported-outcomes-measurement-information-system-promis-and-rapid3-in-rheumatoid-arthritis-patients-starting-a-new-biologic-dmard/. Accessed .« Back to 2019 ACR/ARP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/correlation-between-patient-reported-outcomes-measurement-information-system-promis-and-rapid3-in-rheumatoid-arthritis-patients-starting-a-new-biologic-dmard/