Session Information
Session Type: Abstract Submissions (ACR)
Background/Purpose:
Alpha-enolase (ENO1) is a multifunctional glycolytic enzyme ubiquitously expressed in the cytoplasm. Citrullinated ENO1 is reported to be a candidate autoantigen in rheumatoid arthritis (RA), yet its specific biologic function remains unknown. Histologic analysis indicates that ENO1 is expressed in fibroblast-like synoviocytes (FLS), monocytes, and endothelial cells in the synovium. In pro-inflammatory conditions, ENO1 is translocated to the cell surface where it activates plasminogen. Its expression in monocytes mediates migration of the cells into inflamed lung tissues in animal models. Our aim was to investigate the role of surface-expressed ENO1 in RA FLS, the key constituent of pannus in RA synovium.
Methods :
FLS from RA synovial tissues were isolated and cultured in vitro. ENO1 expression on the cell surface was assessed by confocal microscopy. Cell surface-expressed ENO1 was treated with a mouse anti-human ENO1-stimulating monoclonal antibody as well as an isotype control. Scratch test of cultured FLS and transwell experiments under platelet-derived growth factor (PDGF) were performed to assess FLS migration. Cytoskeletal rearrangement was analyzed after staining FLS with an anti-fillagrin antibody. Fluo-4 fluorophore was used for calcium-flux assays directly on discs plated with FLS.
Results :
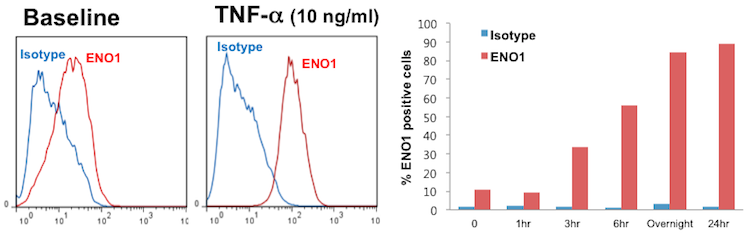
Cell surface ENO1 expression was low in (5~6 passages) cultured FLS under normal condtions. However, overnight TNF-alpha (as low as 0.1 ng/ml) treatment induced ENO1 translocation to the cell surface, which peaked at 24 hours (figure). Stimulation of cell surface-expressed ENO1 induced faster repopulation of FLS in the scratch test assay, and increased PDGF-induced transwell migration. Cytoplasmic actin filament rearrangement in FLS was markedly enhanced with ENO1 stimulation. Moreover, ENO1 stimulation induced a positive calcium flux response in FLS under intravital confocal microscopy.
Conclusion:
Translocation of cytoplasmic ENO1 to the cell surface in RA FLS is potentiated by TNF-alpha. Our results indicate that ENO1 can contribute to migration of RA FLS, especially under the pro-inflammatory milieu as in the RA synovium.
Disclosure:
K. Shin,
None;
J. A. Park,
None;
S. Bae,
None;
J. S. Kang,
None;
Y. W. Song,
None.
« Back to 2012 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/alpha-enolase-facilitates-migration-of-fibroblast-like-synoviocytes-in-rheumatoid-arthritis/