Session Information
Session Type: ACR Poster Session A
Session Time: 9:00AM-11:00AM
Background/Purpose:
Osteoarthritis (OA) is a chronic and progressive degenerative disease. While management of OA should be directed to long-term control of symptoms and joint structure changes, existing pharmacological agents are mostly studied for their effects on symptoms for short-term periods. This is the first systematic review and meta-analysis investigating the effects of available medications over long-term treatment courses on symptoms and structure in knee OA.Methods:
The Cochrane Central Register of Controlled Trials (CENTRAL), PubMed, Embase, Scopus and Web of Science were searched for randomized controlled trials (RCTs) of pharmacological interventions in knee OA published until February 29, 2016. Reference lists of retrieved articles were also screened for additional trials. Only RCTs with treatment/follow-up of at least one year were eligible. The primary outcome was knee OA pain change from baseline to the endpoint (≥ 12 months) on a validated scale. Secondary outcomes were changes in physical function and joint structure expressed as radiologic medial tibiofemoral joint space narrowing (JSN). We performed a random-effects network meta-analysis within a Bayesian framework. Imputation methods for mean changes and variability measures were adopted to include papers with incomplete data. Quality of evidence was rated based on the GRADE approach.Results:
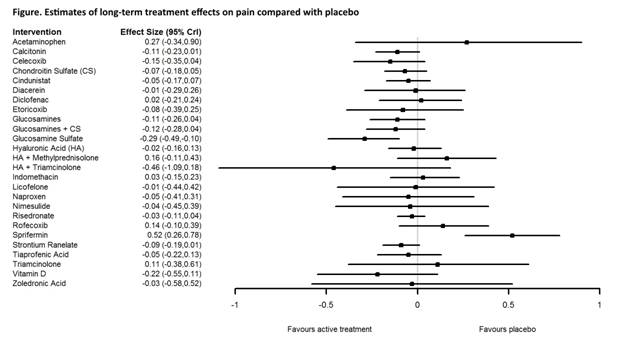
Out of 5992 articles for RCTs of drug therapy in knee OA, 38 RCTs involving 18833 patients met the long-term eligibility criteria and included virtually all available pharmacological intervention categories: 27 interventions including placebo for pain (Figure), 13 for physical function and 17 for JSN, with trial duration ranging between 1 and 3 years. There was no evidence of efficacy for most interventions vs placebo, with the exception of prescription glucosamine sulfate that was significant on pain (Figure) and physical function, with a Glass’ Delta Effect Size (ES) of -0.29 [95% credibility interval: -0.49; -0.10] and -0.32 [-0.52, -0.12], respectively, and high quality of evidence rated by GRADE. Glucosamine sulfate, chondroitin sulfate and strontium ranelate were the only interventions able to significantly reduce radiologic JSN (ES 0.42 [0.20; 0.64], 0.20 [0.08; 0.31] and 0.20 [0.06; 0.35], respectively).Conclusion:
This network meta-analysis shows no evidence of efficacy in the long-term management (at least one year) of knee OA for available medications including NSAIDs, corticosteroids, acetaminophen, putative disease-modifiers, most bone acting agents or Slow Acting Drugs in OA. The only exception is prescription glucosamine sulfate, that is consistently effective on symptoms and joint structure changes, while chondroitin sulfate and strontium ranelate are effective only on structure. Additional long-term RCTs of available and new medications are needed in OA.
Disclosure: D. Gregori, Rottapharm Biotech, 5; G. Giacovelli, Rottapharm Biotech, 3; C. Minto, Rottapharm Biotech, 5; B. Barbetta, Rottapharm Biotech, 3; F. Gualtieri, Rottapharm Biotech, 3; D. Azzolina, Rottapharm Biotech, 5; P. Vaghi, Rottapharm Biotech, 3; L. C. Rovati, Rottapharm Biotech, 3.
To cite this abstract in AMA style:
Gregori D, Giacovelli G, Minto C, Barbetta B, Gualtieri F, Azzolina D, Vaghi P, Rovati LC. A Systematic Review and Network Meta-Analysis of Long-Term Trials of Pharmacological Treatments in Knee Osteoarthritis [abstract]. Arthritis Rheumatol. 2016; 68 (suppl 10). https://acrabstracts.org/abstract/a-systematic-review-and-network-meta-analysis-of-long-term-trials-of-pharmacological-treatments-in-knee-osteoarthritis/. Accessed .« Back to 2016 ACR/ARHP Annual Meeting
ACR Meeting Abstracts - https://acrabstracts.org/abstract/a-systematic-review-and-network-meta-analysis-of-long-term-trials-of-pharmacological-treatments-in-knee-osteoarthritis/