Session Information
Session Type: Poster Session D
Session Time: 1:00PM-3:00PM
Background/Purpose: Guselkumab (GUS) is associated with robust and sustained improvement in PsA signs and symptoms in subgroups of patients (pts) pooled from the phase 3 DISCOVER-1 and DISCOVER-2 (D2) trials, across a variety of baseline (BL) pt characteristics through 11, and 2 years (D2 only)2. In this post-hoc analysis using D2 data, we evaluated the efficacy of GUS in inducing long-term (W100) stringent disease control in Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA)-recommended domains across BL characteristics.
Methods: D2 enrolled biologic-naïve adults with active PsA defined as ≥5 swollen and ≥5 tender joint counts (SJC; TJC) and CRP ≥0.6 mg/dL. 739 pts were randomized (1:1:1) and treated with GUS 100 mg Q4W (n=245); GUS 100 mg at W0, W4, then Q8W (n=248); or placebo (PBO; n=246) with crossover to GUS 100 mg Q4W at W24. In this analysis, only GUS-randomized pts were included (n=493). Achievement of the following outcomes at W100 was assessed: minimal disease activity (MDA), ACR 50% improvement (ACR50), ACR70, Investigator’s Global Assessment score of 0 [clear skin] (IGA 0), Psoriatic Arthritis Disease Activity Score – low disease activity (PASDAS LDA), resolution of enthesitis and dactylitis, Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F) response (≥4-point improvement), and Health Assessment Quality Disease Index (HAQ-DI) response (≥0.35-point improvement). BL characteristics of interest were pt sex, baseline BMI, SJC, TJC, PsA duration, CRP, % body surface area (BSA) with psoriasis, and PASI score, and use of conventional synthetic (cs) DMARDs and methotrexate (MTX). Non-responder imputation was used for missing categorical response data.
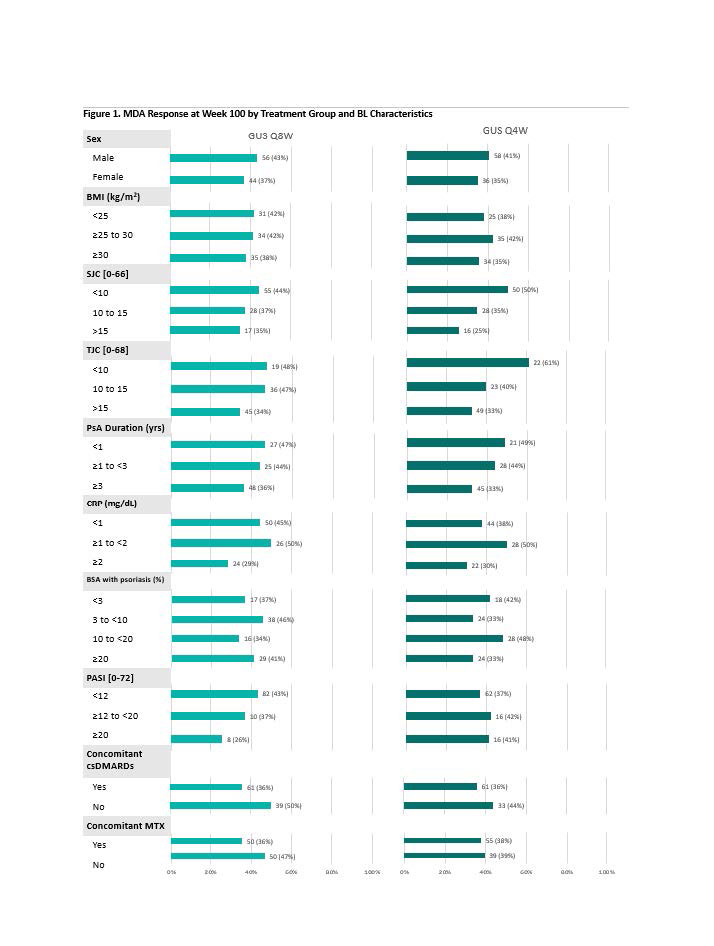
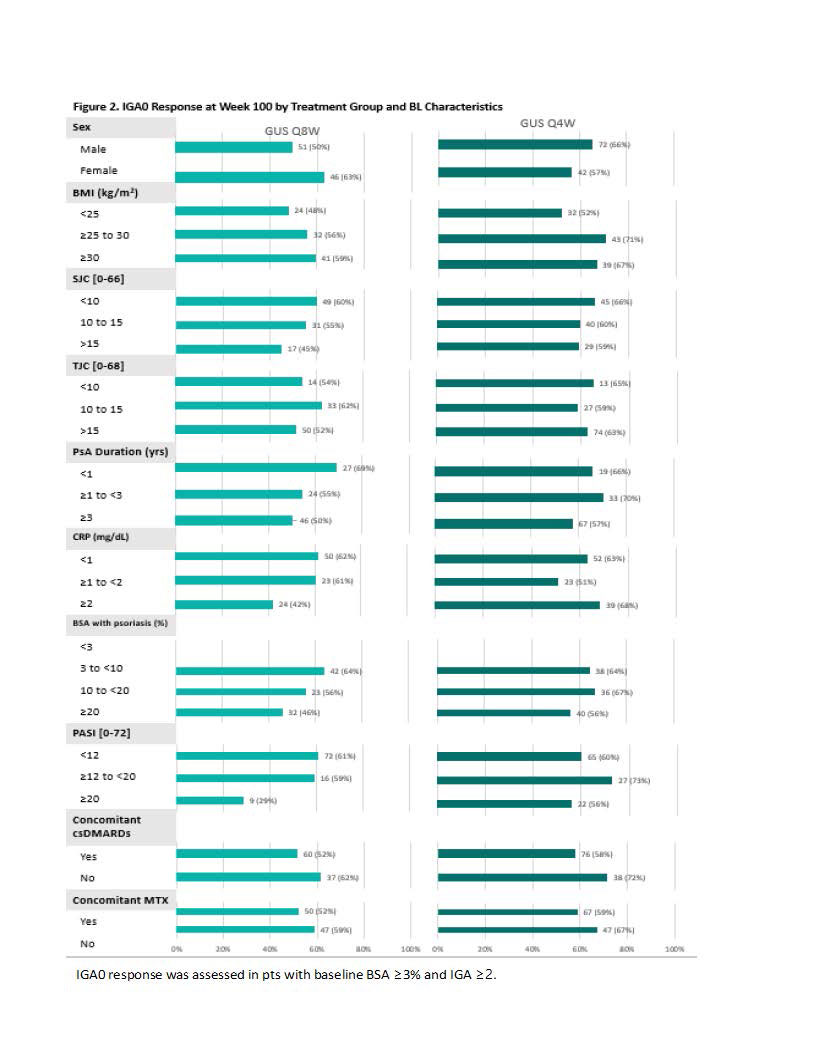
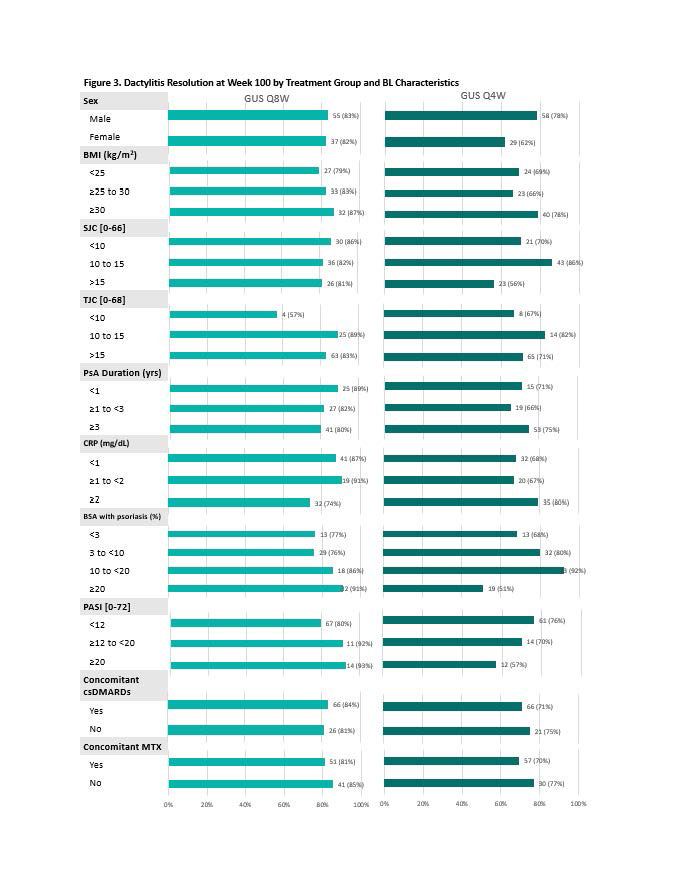
Results: 442 (90%) GUS-randomized pts completed study treatment through W100. With few exceptions, achievement of MDA response (Fig 1), IGA 0 (Fig 2), and resolution of dactylitis (Fig 3) at W100 was demonstrated across a variety of baseline pt characteristics, without consistent differences in proportion of responders across pt subgroups of adequate sample size or between GUS dosing regimens. Similar trends were observed for achievement of ACR50, ACR70, PASDAS LDA, enthesitis resolution, FACIT-F response, and HAQ-DI response.
Conclusion: Irrespective of dosing regimen, treatment with GUS resulted in sustained achievement of several stringent endpoints spanning key GRAPPA-recommended domains through 2 years across a variety of BL demographic and disease characteristics. These results further support the long-term efficacy of GUS across the full spectrum of PsA disease domains and diverse PsA populations.
References:
- Ritchlin CT. et al. RMD Open. 2022 Mar;8(1):e002195.
- Ritchlin CT. et al. Abstract N°AB0526. EULAR 2021.
To cite this abstract in AMA style:
Ritchlin C, Mease P, Boehncke W, Tesser J, Chakravarty S, Rampakakis E, Shawi M, Schiopu E, Merola J, McInnes I, Deodhar A. Stringent Disease Activity Control at 2 Years Across Psoriatic Arthritis Domains Irrespective of Baseline Characteristics in Patients Treated with Guselkumab: Post Hoc Analysis of a Phase 3, Randomized, Double-blind, Placebo-controlled Study [abstract]. Arthritis Rheumatol. 2022; 74 (suppl 9). https://acrabstracts.org/abstract/stringent-disease-activity-control-at-2-years-across-psoriatic-arthritis-domains-irrespective-of-baseline-characteristics-in-patients-treated-with-guselkumab-post-hoc-analysis-of-a-phase-3-randomize/. Accessed .« Back to ACR Convergence 2022
ACR Meeting Abstracts - https://acrabstracts.org/abstract/stringent-disease-activity-control-at-2-years-across-psoriatic-arthritis-domains-irrespective-of-baseline-characteristics-in-patients-treated-with-guselkumab-post-hoc-analysis-of-a-phase-3-randomize/