Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose: Chronic nonbacterial osteomyelitis (CNO) is a debilitating autoinflammatory condition leading to destructive lesions within the bone. CNO can cause significant pain, disability, fractures, and/or long-term deformities. NSAIDs are typically used as first line treatment, while TNF inhibitors (TNFi) are commonly used as second- or third-line treatment. After achieving clinical remission, the optimal duration of treatment is not known. Herein, we explore two-year disease outcomes following discontinuation of either NSAIDs or TNFi therapy in children with CNO.
Methods: We included patients who were diagnosed with CNO before age 18 and enrolled in the CHronic nonbacterial Osteomyelitis International Registry (CHOIR) in the United States. Patients were included if they had achieved clinical remission after receiving NSAID monotherapy or TNFi. Clinical remission was identified when “well-controlled disease” was recorded as the reason for medication discontinuation. Patients that had NSAIDs discontinued after achieving clinical remission between 3-24 months, or TNFi after 6-48 months, were included. Children with inflammatory bowel disease, intolerance of medications, or in the case of the NSAID group, exposure to any second-line treatments or glucocorticoids were excluded. Disease flare was defined as a new spinal or sacroiliac lesion on MRI, clinical arthritis, or an increase in pain or lesion size. Mann-Whitney U test or Fisher’s exact test were performed for each cohort and Kaplan Meier analysis was performed to compare the flare-free probability between short and long treatment duration subgroups.
Results: Within CHOIR, 35 patients achieved clinical disease remission following NSAID monotherapy prior to medication discontinuation (n=10 for 3-9 months [short duration], n=17 for 9-24 months [long duration], 8 excluded for duration < 3 or >24 months). Baseline characteristics were similar between the short and long duration groups (Table 1). During the 2- year follow-up, approximately 70% of patients in the short-duration group and 40% in the long-duration group experienced a disease flare, though this was not statistically significant (p = 0.38, Figure 1A). In the TNFi cohort, 26 patients achieved clinical remission and stopped TNFi within 48 months (n=10 for 3-18 months [short duration] versus n=16 for 18-48 months [long duration]). Distribution of spinal lesions, sacroiliitis, psoriasis, concurrent use of csDMARD, or bisphosphonate were similar between the groups (Table 2). There was no significant difference in disease flares between the short and long-duration groups, with approximately 40% of patients maintaining disease remission during the average 2-year off-medication observation periods (p=0.46, Figure 1B).
Conclusion: There was no statistically significant difference in sustained off-medication remission from either NSAID- or TNFi-responders in this limited dataset. Extended TNFi treatment did not appear to alter the risk of disease flare. Larger studies are necessary to confirm these findings.
Figure 1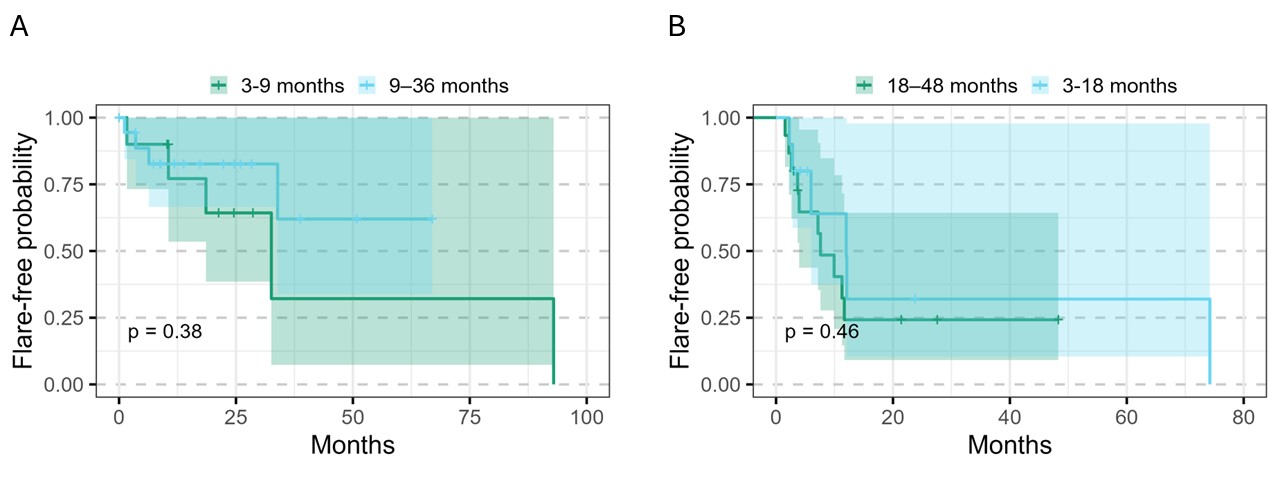 Figure 1. Kaplan-Meier curve of the flare rates from two groups over time after discontinuation of NSAIDs (A) or TNFi (B)
Figure 1. Kaplan-Meier curve of the flare rates from two groups over time after discontinuation of NSAIDs (A) or TNFi (B)
Table 1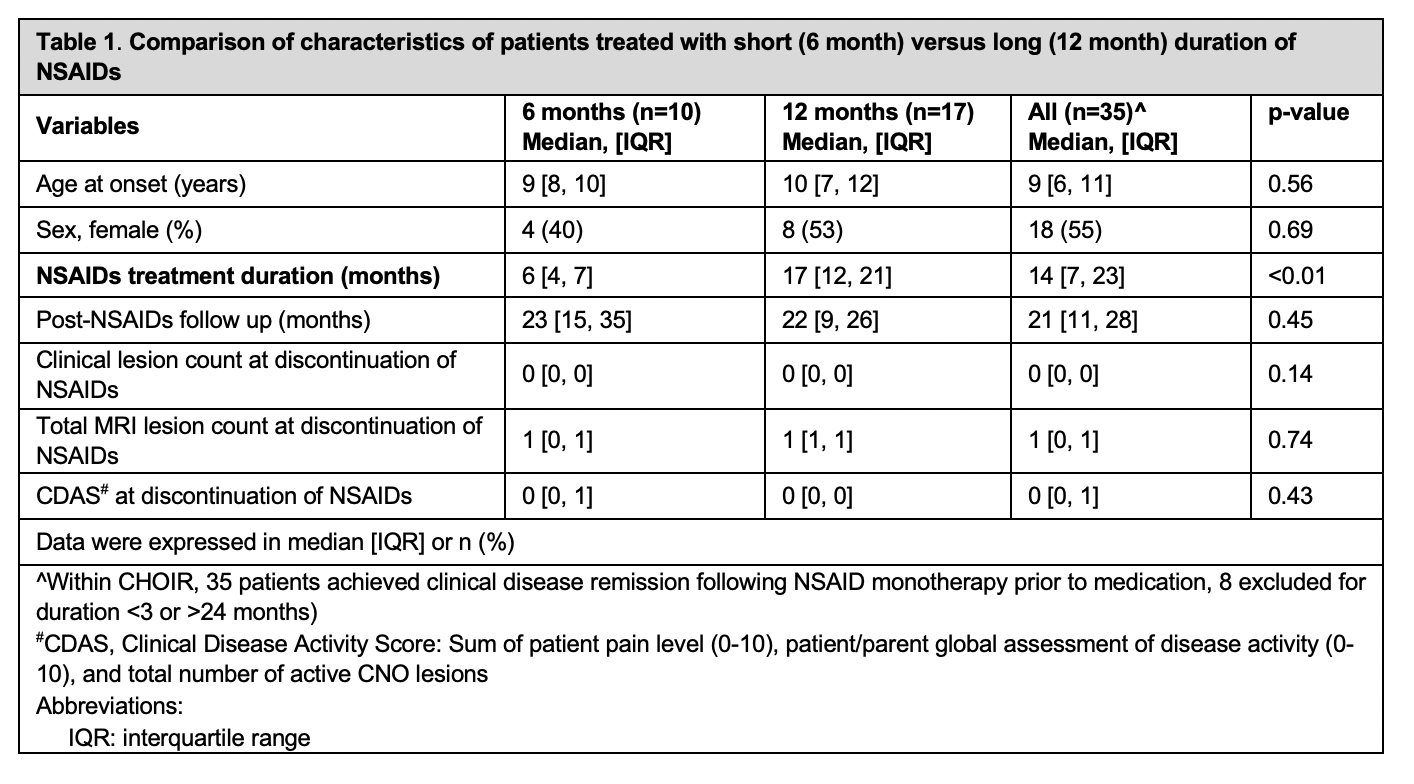 Table 1. Comparison of characteristics of patients treated with short (6 month) versus long (12 month) duration of NSAIDs
Table 1. Comparison of characteristics of patients treated with short (6 month) versus long (12 month) duration of NSAIDs
Table 2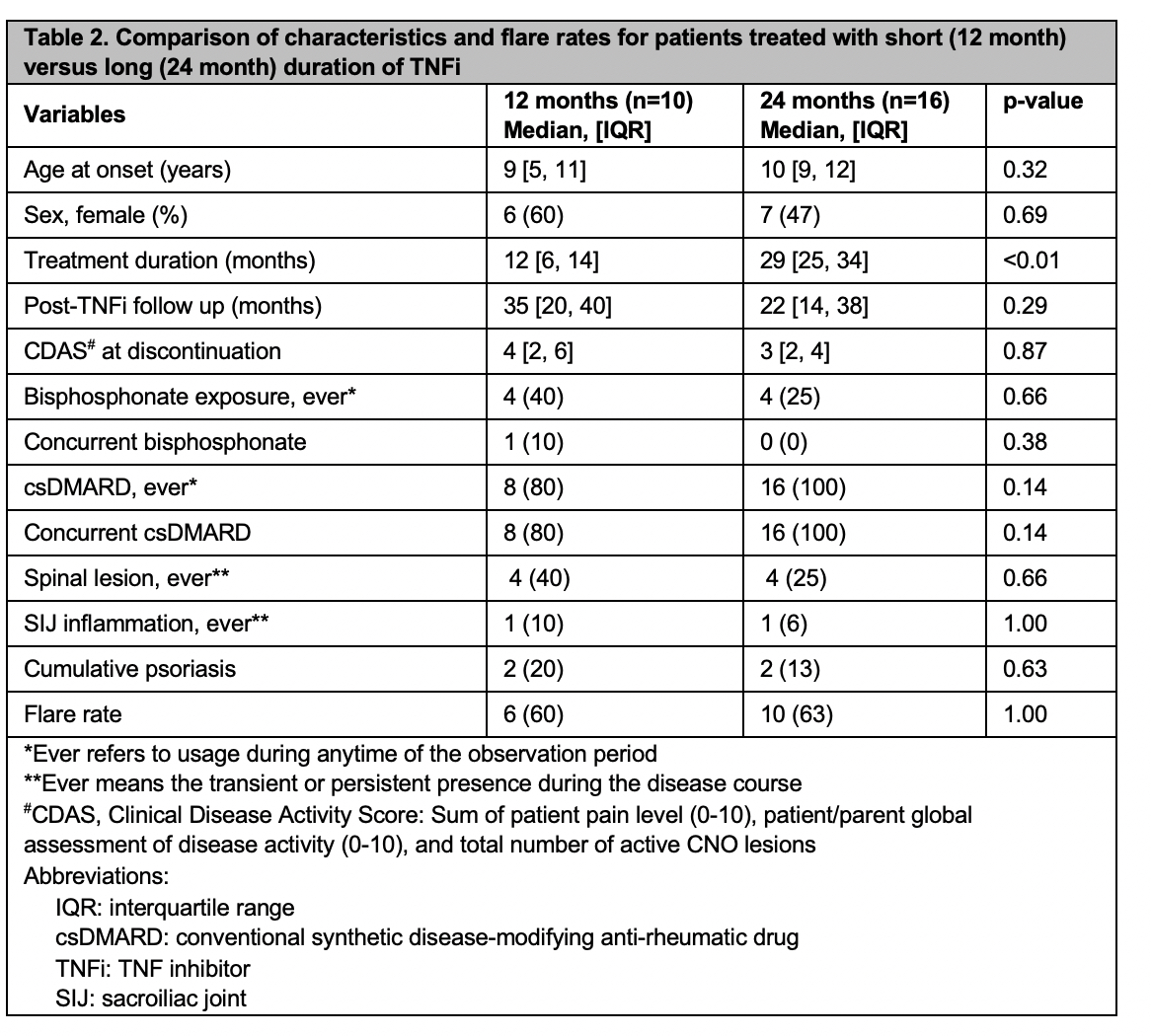 Table 2. Comparison of characteristics and flare rates for patients treated with short (12 month) versus long (24 month) duration of TNFi
Table 2. Comparison of characteristics and flare rates for patients treated with short (12 month) versus long (24 month) duration of TNFi
To cite this abstract in AMA style:
Moussouras N, Wang X, Rosenwasser N, Muse I, Chen H, Wu E, Oliver M, Lapidus S, Liau A, Klein A, Wilson A, Deng E, Li T, Nguyen M, Morden J, Yasin S, Lenert A, Stern S, Huang B, Kovalick L, Balay-Dustrude E, Wong S, Thomason J, Onel K, Dedeoglu F, Ferguson P, Zhao Y. Pediatric chronic nonbacterial osteomyelitis outcomes after NSAID or TNF inhibitor discontinuation, a registry cohort with two year follow up data [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/pediatric-chronic-nonbacterial-osteomyelitis-outcomes-after-nsaid-or-tnf-inhibitor-discontinuation-a-registry-cohort-with-two-year-follow-up-data/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/pediatric-chronic-nonbacterial-osteomyelitis-outcomes-after-nsaid-or-tnf-inhibitor-discontinuation-a-registry-cohort-with-two-year-follow-up-data/
