Session Information
Session Time: 4:37PM-4:42PM
Background/Purpose: There are limited studies exploring the pain experience, both report of pain as well as treatment, among patients with childhood onset systemic lupus erythematosus (cSLE).
Methods: Retrospective cohort study of patients with cSLE enrolled between March 2017 to February 2025. Inclusion criteria for subjects enrolled in the CARRA Registry include: 1) diagnosis of cSLE at < 18 years; 2) enrollment within two years of cSLE diagnosis or at the time of a flare of lupus nephritis; and 3) enrollment prior to 21 years of age. Self-report of pain and treatment data were collected at baseline and every 6 months up to 24 months of follow-up. Inclusion criteria in this study cohort include participants who had pain measured at the baseline visit and at least 1 follow-up visit in the 24 month follow-up period. Pain intensity was assessed using a visual analogue scale from 0 (no pain) to 10 (worse possible pain). If a participant had pain intensity ≥ 1, a Patient-Reported Outcomes Measurement Information System (PROMIS) Pain Interference assessment was completed. Provider-reported pain medication data was added to the CARRA SLE Registry in 2020. Data were summarized using descriptive statistics.
Results: 833 participants were included this analysis. Baseline sociodemographic and disease characteristics are summarized in Table 1. Participants had a median age of enrollment of 15 years (13.00, 16.85), median time from diagnosis to enrollment of 3.21 (1.05, 8.95) months and median SLEDAI at enrollment of 6.00 (2.00, 12.00). Pain measures at baseline and at every 6 month visits are shown in Table 2. Median pain intensity score was 2 (0, 4) at baseline and 1 (0, 3) at the 24 month follow-up. Pain interference scores suggest that at baseline and 6 months, participants experience similar pain interference as the average person in the general population 48.40 (34.00, 58.50); pain interference scores decreased to 42.70 (34.00, 55.70) at 24 months. There was no significant difference in baseline pain scores between by sex. At the baseline visit there was a significant difference in pain intensity (2 (0,5) vs 1 (0,4), p= 0.0030) and pain interference (50.6 (34.0,59.5) vs 45.8 (34.0,56.0), p=0.0069) for participants with and without arthritis, respectively.
Pain medication data was available for 409/833 (49.1%) of participants at baseline. As summarized in Table 3, non-steroidal anti-inflammatory medications were the most commonly prescribed medications for cSLE related pain. 7.6% of participants received opioids prior to or at the baseline visit with a decrease in the frequency of opioid, and other pain medication prescription, at subsequent follow-up visits.
Conclusion: This is one of the first studies to describe the pain experience and treatment of pain among a diverse cohort of cSLE patients followed over a 24 month period. Future studies will further examine the relationships between pain with disease activity, specific SLE manifestations and treatment.
Table 1: Baseline characteristics of childhood-onset systemic lupus erythematosus participants in the CARRA registry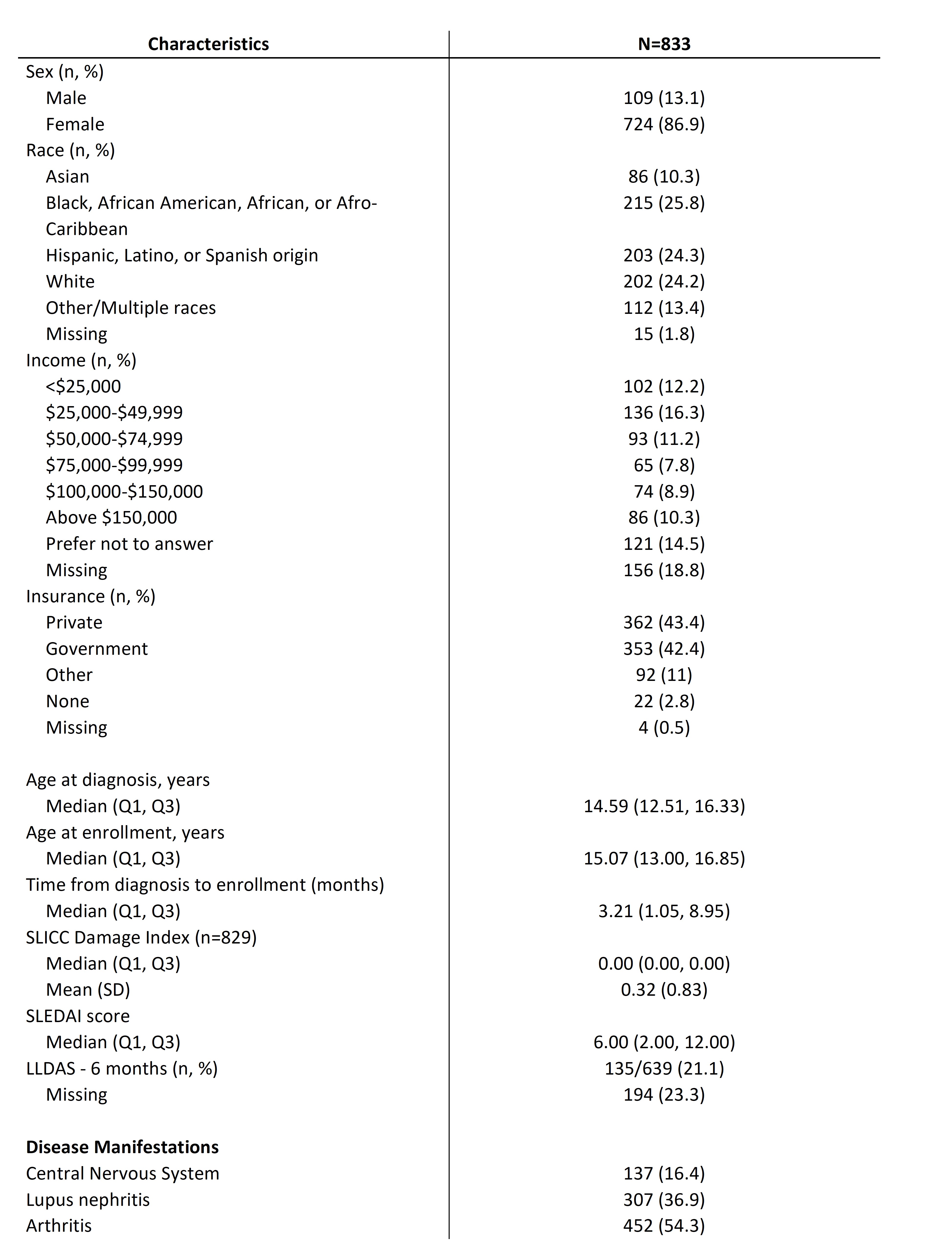
Table 2. Pain measures by visit in the CARRA childhood-onset Systemic Lupus Erythematosus registry. 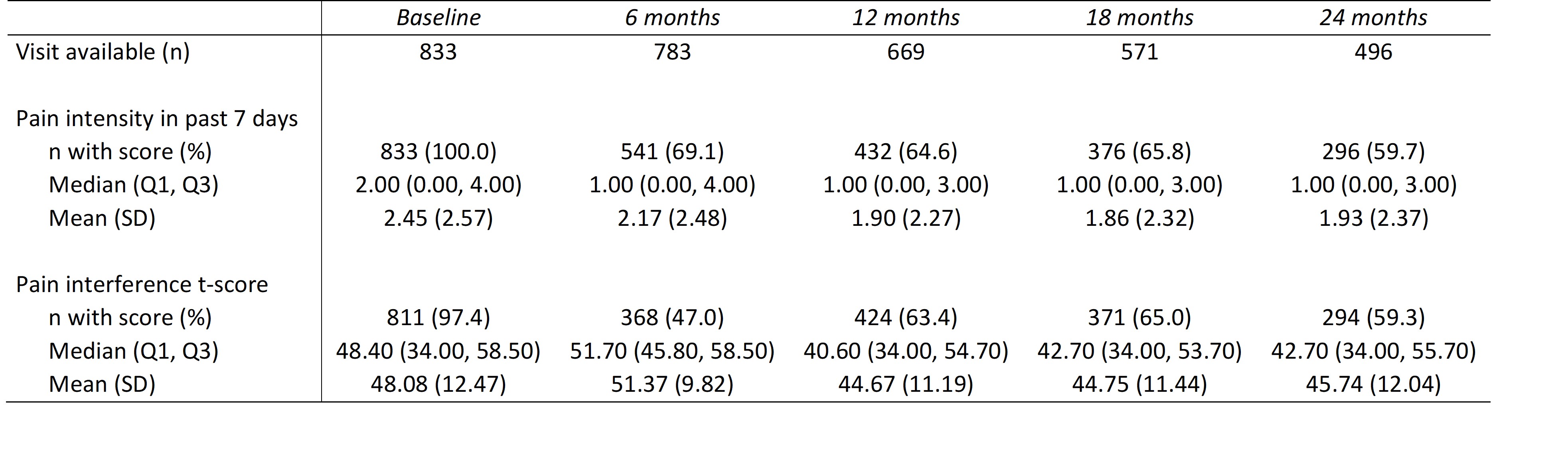
Table 3. Pain medication use in the CARRA childhood-onset Systemic Lupus Erythematosus registry. 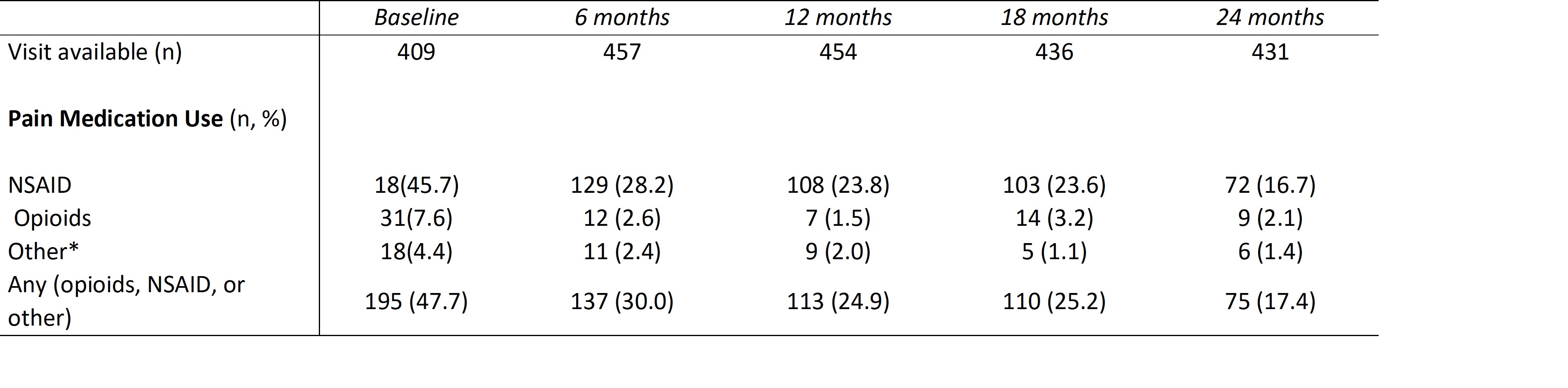 *Other includes gabapentin, pregabalin
*Other includes gabapentin, pregabalin
To cite this abstract in AMA style:
Hersh A, Shrader P, Dennos A, Knight A, Ogbu E, von Scheven E, Schanberg L, Son M. Pain Experience and Treatment in the Childhood Arthritis and Rheumatology Research Alliance (CARRA) Systemic Lupus Erythematosus Registry [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/pain-experience-and-treatment-in-the-childhood-arthritis-and-rheumatology-research-alliance-carra-systemic-lupus-erythematosus-registry/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/pain-experience-and-treatment-in-the-childhood-arthritis-and-rheumatology-research-alliance-carra-systemic-lupus-erythematosus-registry/
