Session Information
Session Time: 6:00PM-7:00PM
Background/Purpose: The REGENCY trial (NCT04221477) demonstrated superiority of obinutuzumab (OBI) plus standard therapy (+ST) vs placebo (PBO) +ST in achieving complete renal response (CRR) at Week 76 (W76) in adults with active lupus nephritis (LN). It was postulated that OBI+ST would yield greater rates of histologic remission and kidney tissue-level B-cell depletion at W76 than PBO+ST, which would portend more favorable long-term kidney outcomes, such as reduced LN flare risk and preserved kidney function. These exploratory analyses aimed to evaluate histologic remission and kidney tissue-level B-cell depletion at W76 in patients treated with OBI+ST vs PBO+ST.
Methods: Paired baseline and W76 kidney biopsies from REGENCY participants with biopsy-proven proliferative LN were analyzed. Histologic analysis: 64 biopsies (32 OBI+ST, 32 PBO+ST) were evaluated using the 2018 International Society of Nephrology/Renal Pathology Society LN classification, along with the National Institute of Health activity (AI) and chronicity indices. The proportion of patients achieving histologic or near-histologic remission (AI=0 or ≤1) was determined. B-cell analysis: 29 participants (14 OBI+ST, 15 PBO+ST) were assessed. CD79a+/CD138− B cells were quantified by immunofluorescence microscopy and digital whole-slide analysis. Changes in B-cell counts at W76 were compared using an ANCOVA model, adjusting for baseline B-cell counts and stratification factors.
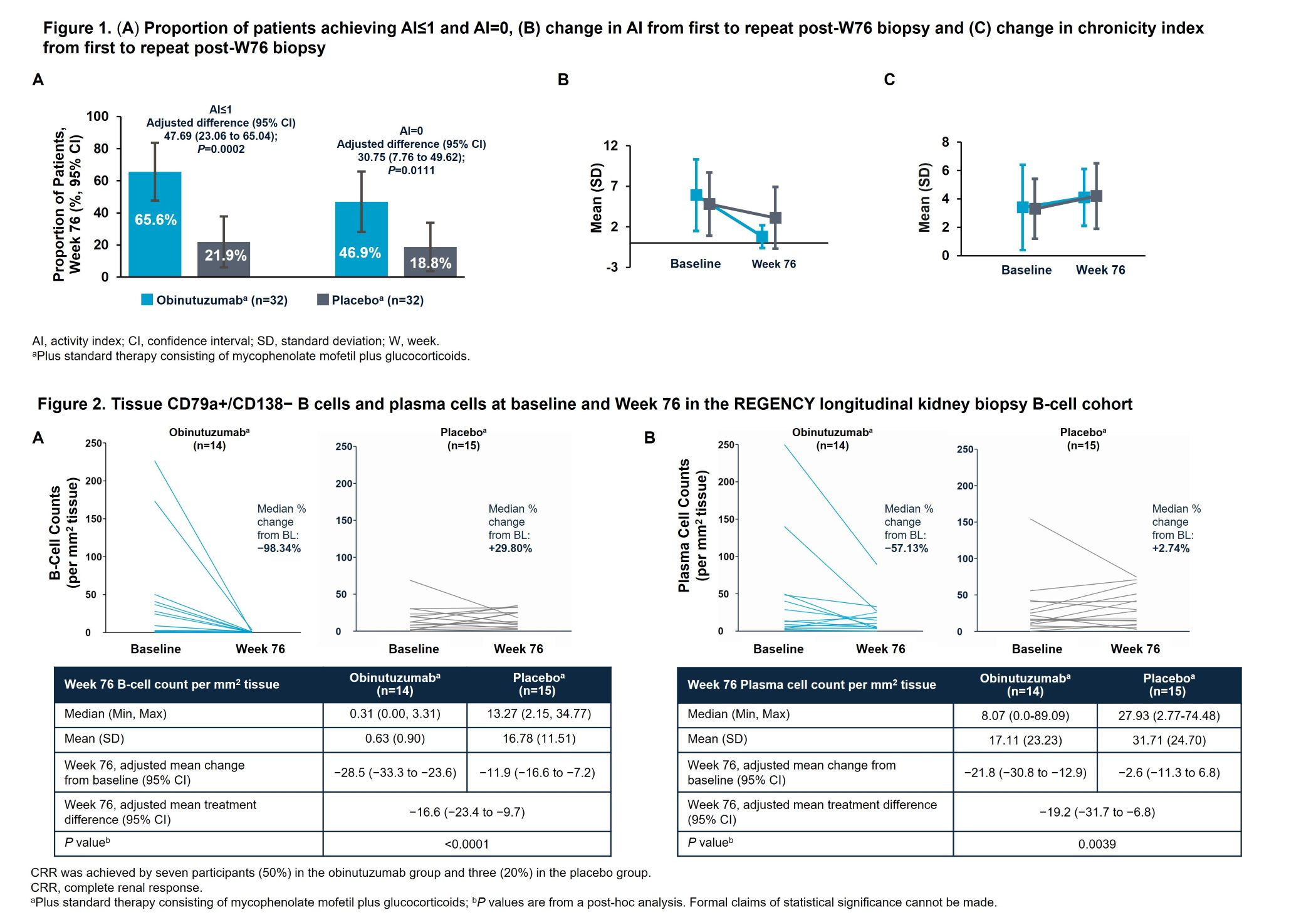
Results: Baseline characteristics were balanced, despite higher tissue B-cell levels in the OBI+ST group. At W76, significantly more patients achieved AI=0 or ≤1 with OBI+ST vs PBO+ST (Figure 1). Among patients not achieving CRR, 52.6% (10/19) in the OBI+ST group had an AI=0 at W76, vs 8.3% (2/24) in the PBO+ST group. Nearly every patient in the OBI+ST group had their kidney tissue B-cell count drop substantially, approaching zero, by W76 (Figure 2). The adjusted mean change in B-cell counts from baseline to W76 was −28.5 (95% CI, −33.3 to −23.6) for OBI+ST vs −11.9 (95% CI, −16.6 to −7.2) for PBO+ST, a significant difference of −16.6 (95% CI, −23.4 to −9.7; P< 0.0001).
Conclusion: In the largest longitudinal kidney biopsy cohort ever reported for a registrational LN clinical trial, significantly more patients achieved complete or near-complete histologic remission with OBI+ST vs PBO+ST. This is the first demonstration of deep kidney tissue B-cell depletion by any anti-CD20 agent, in any glomerular disease. Obinutuzumab’s potent B-cell clearance from kidney tissue may drive kidney function improvement and LN flare reduction. These findings support assessment of histologic outcomes in future LN trials and highlight a potential mechanism for obinutuzumab in preserving long-term kidney health.
To cite this abstract in AMA style:
Rovin B, Martins E, Austin C, Raghu H, Chan C, Chang P, Garg J, Alberton V, Santiago M, Aroca-Martínez G, Palazuelos F, Baczkowska T, Alfaro J, Ravelo-Hernandez J, Furie R, Pinto L, Albeiro E, Larsen C, Yoo B, Pulley J, Thorley A, Schindler T, Omachi T, Pendergraft W, Loflin B, Malvar A. Obinutuzumab Induces Histologic Remission And Deep Kidney Parenchymal B-Cell Depletion In Patients With Lupus Nephritis: Exploratory Analyses From The REGENCY Trial [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/obinutuzumab-induces-histologic-remission-and-deep-kidney-parenchymal-b-cell-depletion-in-patients-with-lupus-nephritis-exploratory-analyses-from-the-regency-trial/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/obinutuzumab-induces-histologic-remission-and-deep-kidney-parenchymal-b-cell-depletion-in-patients-with-lupus-nephritis-exploratory-analyses-from-the-regency-trial/