Session Information
Session Time: 5:11PM-5:16PM
Background/Purpose: Lung disease is a severe and potentially fatal complication of systemic juvenile idiopathic arthritis (SJIA-LD). The Childhood Arthritis and Rheumatology Research Alliance (CARRA) Registry SJIA-LD cohort was established to prospectively determine disease course, treatments, and outcomes. Here, we report 6-month follow-up data and changes in lung disease activity.
Methods: Existing or newly enrolled CARRA Registry patients with SJIA and suspected, probable, or definite SJIA-LD were included in this cohort. In addition to standard Registry data, lung disease specific data was obtained at baseline and at 6 month follow-up visits using a standardized case report form through REDCap Cloud. Biosamples were also collected at baseline and follow-up using CARRA standard Type I kits. KL-6 levels were determined by specific ELISA. This study was approved by the DCRI Reliant IRB and/or IRB of Registry sites, and all patients or parents/guardians provided informed consent for Registry enrollment and biosample collection.
Results: Baseline and follow-up visit data was available for 36 patients from 16 CARRA Registry sites (Table 1). Follow-up visit data was collected at a median of 203 days after baseline (Figure 1; IQR 165-261, range 105-980 days). During follow-up, 16 patients (44.4%) stopped at least one medication. The most commonly discontinued medications were anakinra (5 patients), tocilizumab (3), cyclosporine (2), or infliximab (2). Twelve patients (33.3%) started a new medication, the most common being ruxolitinib (3 patients), tofacitinib (2), emapalumab (2), or MAS825 (1).
While the mean physician global assessment (PGA) did not significantly change from baseline to follow-up (mean 2 vs 1.5, p=0.39), the mean physician global assessment of lung disease (PGALD) significantly decreased (mean 3.3 vs 1.8, p=0.006) (Figure 1). 18 patients (50%) improved the PGALD by ≥2 points while in 2 patients (5.5%) the PGALD worsened by ≥2 points. Similarly, clinical lung disease was judged as somewhat or much better in 13 patients (36.1%), the same in 21 (58.3%), and somewhat worse in 2 (5.5%). 22 patients had repeat chest CT during follow-up period: 7 (31.8%) were judged as somewhat or much better, 14 (63.6%) unchanged, and 1 (4.5%) somewhat worse.
Twenty patients had baseline and follow-up biosamples available for analysis. Median levels of KL-6 were 593 (IQR 237-6975) at baseline and 494 (273-9565) at follow-up. KL-6 levels at baseline and follow-up were highly correlated (R=0.76, p< 0.001) and did not significantly change (Figure 1).
Conclusion: At six-month follow-up, most patients in the CARRA Registry SJIA-LD cohort showed stable- to improved clinical lung disease activity. However, KL-6 levels as a marker of lung injury were largely unchanged. Patients showed frequent medication changes with less use of IL-1/6 inhibitors and initiation of novel medications for SJIA and MAS. Continued enrollment and long-term follow-up is essential to better understand the clinical patterns, morbidity and mortality of SJIA-LD.
Table 1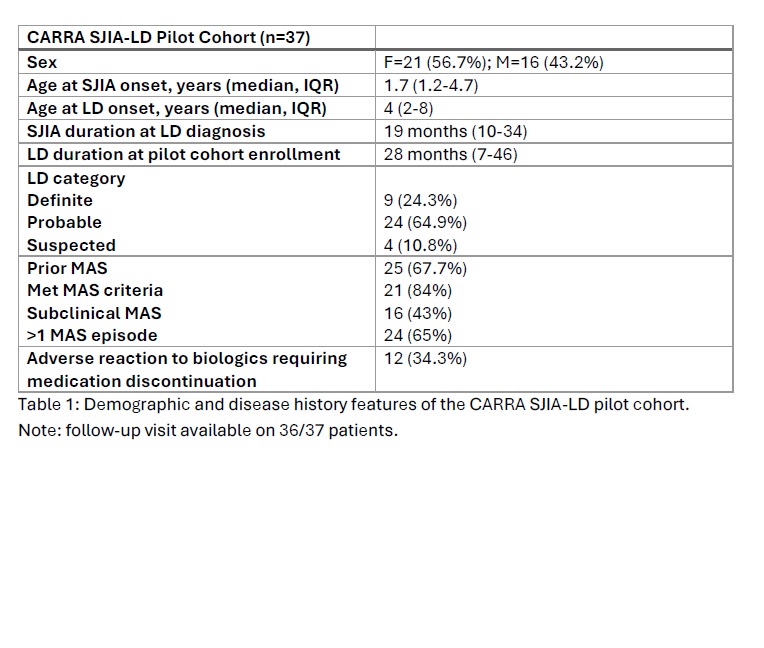 Demographic and disease history features of the CARRA SJIA-LD pilot cohort. Note: follow-up visit available on 36/37 patients.
Demographic and disease history features of the CARRA SJIA-LD pilot cohort. Note: follow-up visit available on 36/37 patients.
Figure 1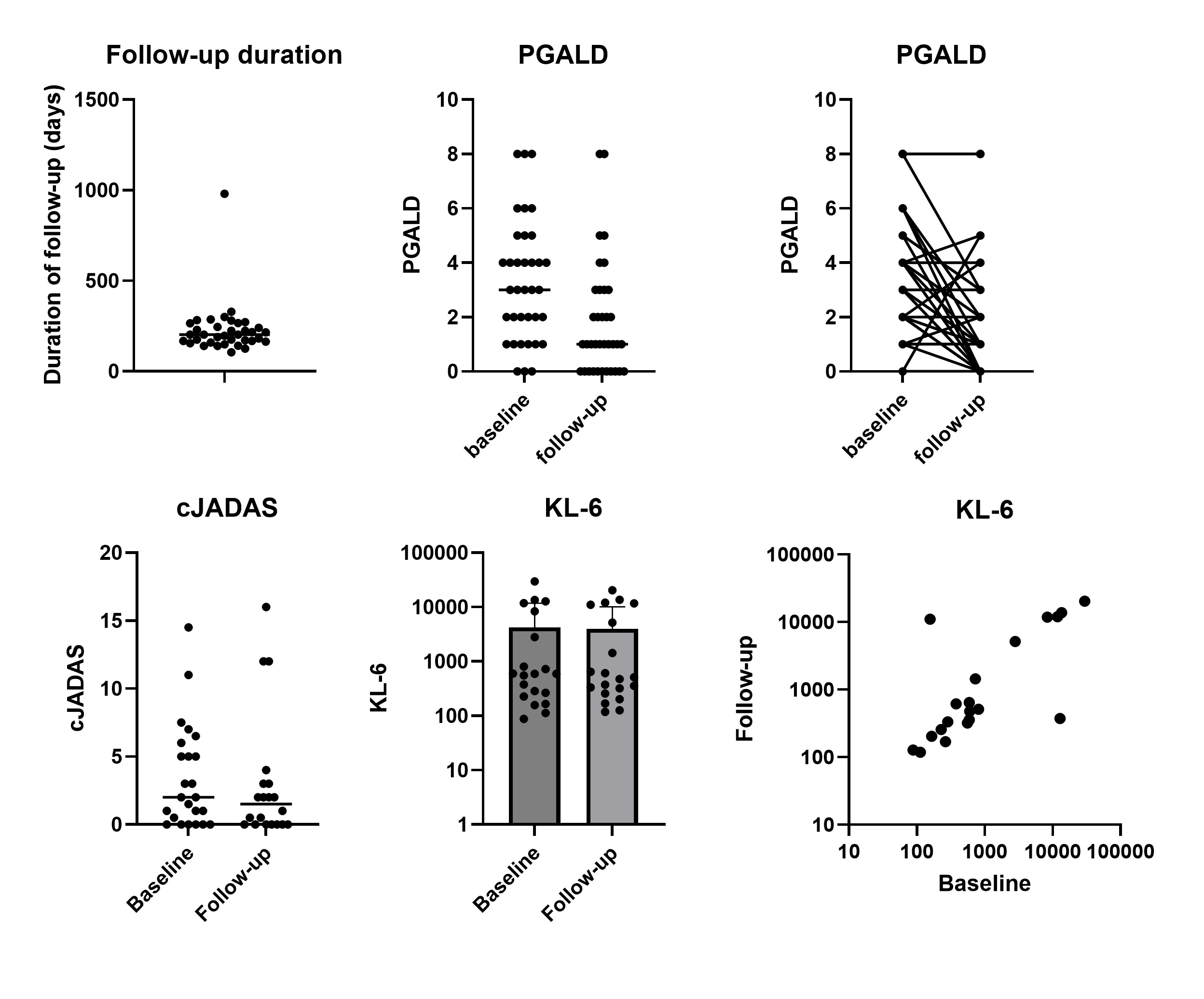 Six-Month Follow-Up of CARRA Registry SJIA-LD Cohort. Top left: duration of follow-up visit from baseline cohort visit. Top middle and right: PGALD at follow-up visit. Bottom left: cJADAS at follow-up visit. Bottom middle and right: KL-6 levels at baseline and follow-up visits.
Six-Month Follow-Up of CARRA Registry SJIA-LD Cohort. Top left: duration of follow-up visit from baseline cohort visit. Top middle and right: PGALD at follow-up visit. Bottom left: cJADAS at follow-up visit. Bottom middle and right: KL-6 levels at baseline and follow-up visits.
To cite this abstract in AMA style:
Schulert G, Rife E, Riordan M, Russell A, Ong M, Eloseily E, Auld L, Kimura Y. Medication Changes and Lung Disease Activity in Systemic JIA: Six-Month Follow-Up of CARRA Registry SJIA-LD Cohort [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/medication-changes-and-lung-disease-activity-in-systemic-jia-six-month-follow-up-of-carra-registry-sjia-ld-cohort/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/medication-changes-and-lung-disease-activity-in-systemic-jia-six-month-follow-up-of-carra-registry-sjia-ld-cohort/
