Session Information
Session Time: 5:29PM-5:34PM
Background/Purpose: Childhood lupus nephritis (cLN) is a life-threatening manifestation of systemic lupus erythematosus and frequently demonstrates incomplete response to standard immunosuppression. The mechanisms driving persistent inflammation remain poorly understood, in part because existing spatial profiling platforms measure RNA, rather than protein expression, of 1,000 or fewer genes. The purpose of this study was to leverage a high-plex, integrated spatial transcriptomics and proteomics platform to map 6,000 genes and 64 proteins across hundreds of thousands of cells from pediatric cLN biopsies. By comparing diseased kidneys to healthy tonsillar immune architecture, we aimed to identify aberrant lymphocyte developmental states and immune circuits that may represent novel therapeutic targets.
Methods: Patients with biopsy-confirmed class III or IV cLN, and a patient who received a nephrectomy for a non-inflammatory indication, were enrolled in a no-contact clinical study approved by the Seattle Children’s Hospital Institutional Review Board (IRB). Subsequently, previous biopsies obtained for clinical diagnostic purposes at or near diagnosis were subject to the CosMx Human Multiomics platform (Bruker). De-identified tonsillar tissue obtained as part of routine clinical care was subject to the same platform. Cell profiles were subject to Insitutype, a Bayesian method for identifying cell types, using the Kidney Precision Medicine Project (KPMP) v1 reference cell types; or subject to unsupervised clustering via PCA and UMAP using the transcriptional data only. Differential gene testing and spatial relationship analyses were performed for lymphocyte cell subclusters that were shared between tonsillar and cLN tissues.
Results: Supervised cell identification of 47,470 kidney cells revealed a large increase in the number of lymphocytes observed in cLN (13.1% of all cells profiled), compared to the non-inflammatory kidney control (0.5% of all cells profiled). Approximately 33% of these highly abundant lupus nephritis lymphocytes were mapped to clusters also observed in the healthy tonsillar tissue. Comparing “tonsillar-like” to “non-tonsillar” B lymphocytes revealed differential gene patterns associated with antigen presentation, specifically upregulation of MHC molecules and CD37, which are critical for T-B cell interactions in germinal centers. Spatial relationships between cell subclusters is ongoing as of the submission of this abstract.
Conclusion: The striking similarities between tonsillar and lupus nephritis lymphocytes suggests that there is aberrant lymph-node like differentiation of B and T cells within the interstitial space in patients with cLN. This is congruent with past reports of tertiary lymphoid structures being observed in autoimmune diseases. The maturation of B and T cells in the kidney suggests that at least some autoreactive T and B cells are continuously generated in situ, rather than being generated solely in dedicated secondary lymphoid follicles. This in situ autoreactive lymphocyte development may be a keystone in the pathogenesis of lupus nephritis, as evidenced by the recent success of deep CD19-directed B cell-depleting therapies.
UMAP of all cells from patients with lupus nephritis, control patients, and healthy tonsillar tissue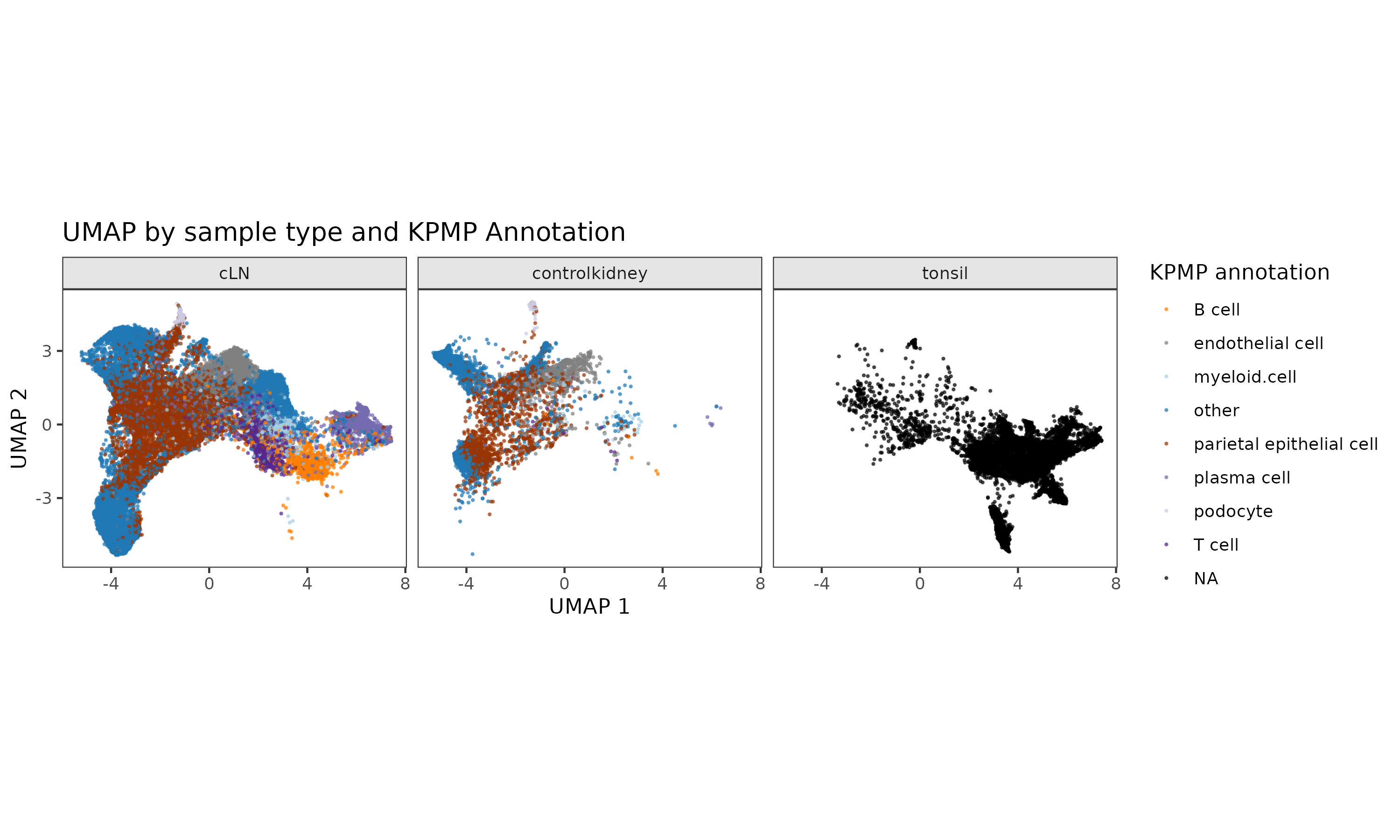 Cells obtained using needle kidney biopsy are colored according to their cell type, as determined using Insitutype, a Bayesian algorithm for supervised cell type identification, using the Kidney Precision Medicine Partnership (KPMP) reference dataset.
Cells obtained using needle kidney biopsy are colored according to their cell type, as determined using Insitutype, a Bayesian algorithm for supervised cell type identification, using the Kidney Precision Medicine Partnership (KPMP) reference dataset.
To cite this abstract in AMA style:
Hasle N, Reed R, Hoglund V, Martin R, Conner M, Jackson S. Integrated Spatial Transcriptomics and Proteomics in Reveals Aberrant Lymphoid Networks in Pediatric Lupus Nephritis [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/integrated-spatial-transcriptomics-and-proteomics-in-reveals-aberrant-lymphoid-networks-in-pediatric-lupus-nephritis/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/integrated-spatial-transcriptomics-and-proteomics-in-reveals-aberrant-lymphoid-networks-in-pediatric-lupus-nephritis/
