Session Information
Session Time: 5:00PM-6:00PM
Background/Purpose: Juvenile dermatomyositis (JDM) is a rare, chronic autoimmune myopathy characterized by muscle inflammation and distinctive skin manifestations. Although corticosteroids and conventional immunosuppressants remain the mainstay of therapy, a subset of patients develop chronic or refractory disease with persistent inflammation, calcinosis, or vasculopathy, requiring escalation to steroid sparing immune modulatory agents. Among the biologic agents, Abatacept and Janus kinase (JAK) inhibitors, are increasingly used among these patients, with the latter being guided by certain phenotypes and myositis specific antibodies (anti-MDA5 and anti-NXP2). Evaluating their real world effectiveness and safety in a tertiary care setting is essential to inform clinical practice and improve outcomes for children with refractory JDM.
Methods:
With IRB approval, we conducted a retrospective chart review of JDM patients at Texas Children’s Hospital from January 2011 to June 2024. We included patients with severe, chronic, or refractory disease treated with JAK inhibitors (tofacitinib, baricitinib, ruxolitinib), abatacept, or other rescue therapies (control group). Extracted data included demographics, clinical features, disease course, and outcomes. Treatment response was based on validated disease activity score (DAS) and adverse events were assessed at 3, 6, and 12 months. Group differences were analyzed using Fisher’s exact test for categorical variables and Kruskal–Wallis for continuous variables. Longitudinal DAS changes were evaluated with multivariable mixed-effects models adjusted for age, gender, race, ethnicity, and time to diagnosis. Statistical significance was set at p< 0.05.
Results: Twenty-three patients were included: seven treated with JAK inhibitors, eight with abatacept, and eight controls. All patients on targeted therapies also received conventional synthetic disease-modifying antirheumatic drugs. All groups showed improvement in disease activity within one year, but only the abatacept group demonstrated a statistically significant reduction in DAS muscle scores. In the JAK inhibitor group, MDA5-positive patients showed a nonsignificant trend toward earlier disease control, likely given the small number of this cohort. IVIG use declined across all groups, suggesting a steroid-sparing effect. No major adverse events occurred in the first year, supporting the short-term safety of both abatacept and JAK inhibitors.
Conclusion:
In this single center retrospective study, abatacept showed greater improvement in muscle disease activity than JAK inhibitors or controls, with no serious short-term safety concerns. These results support abatacept as a promising option for refractory JDM and underscore the need for larger prospective studies. No serious adverse events occurred within one year for either therapy. Although not statistically significant, JAK inhibitors in MDA5-positive patients were associated with earlier remission and fewer relapses.
Baseline Characteristics Between Groups 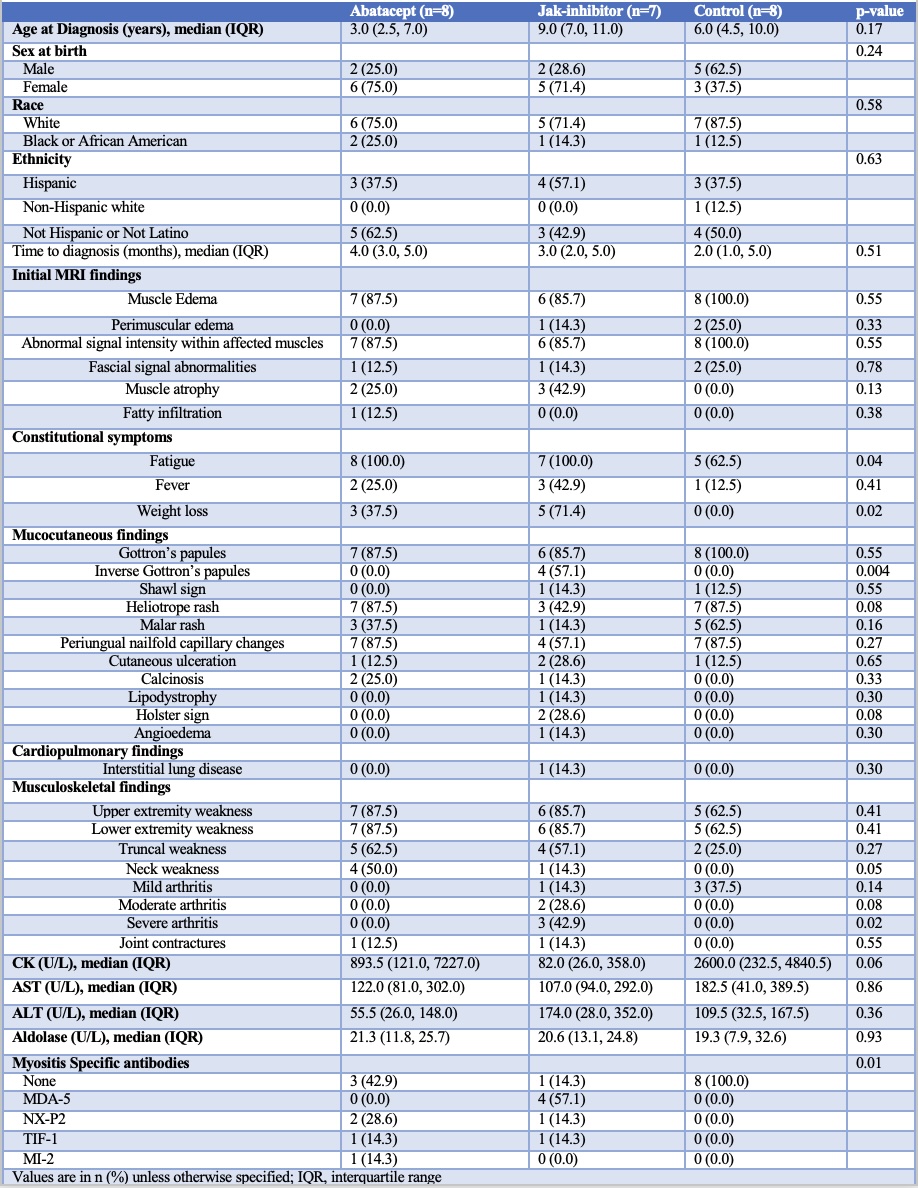
Indications for Initiating JAK Inhibitors vs. Abatacept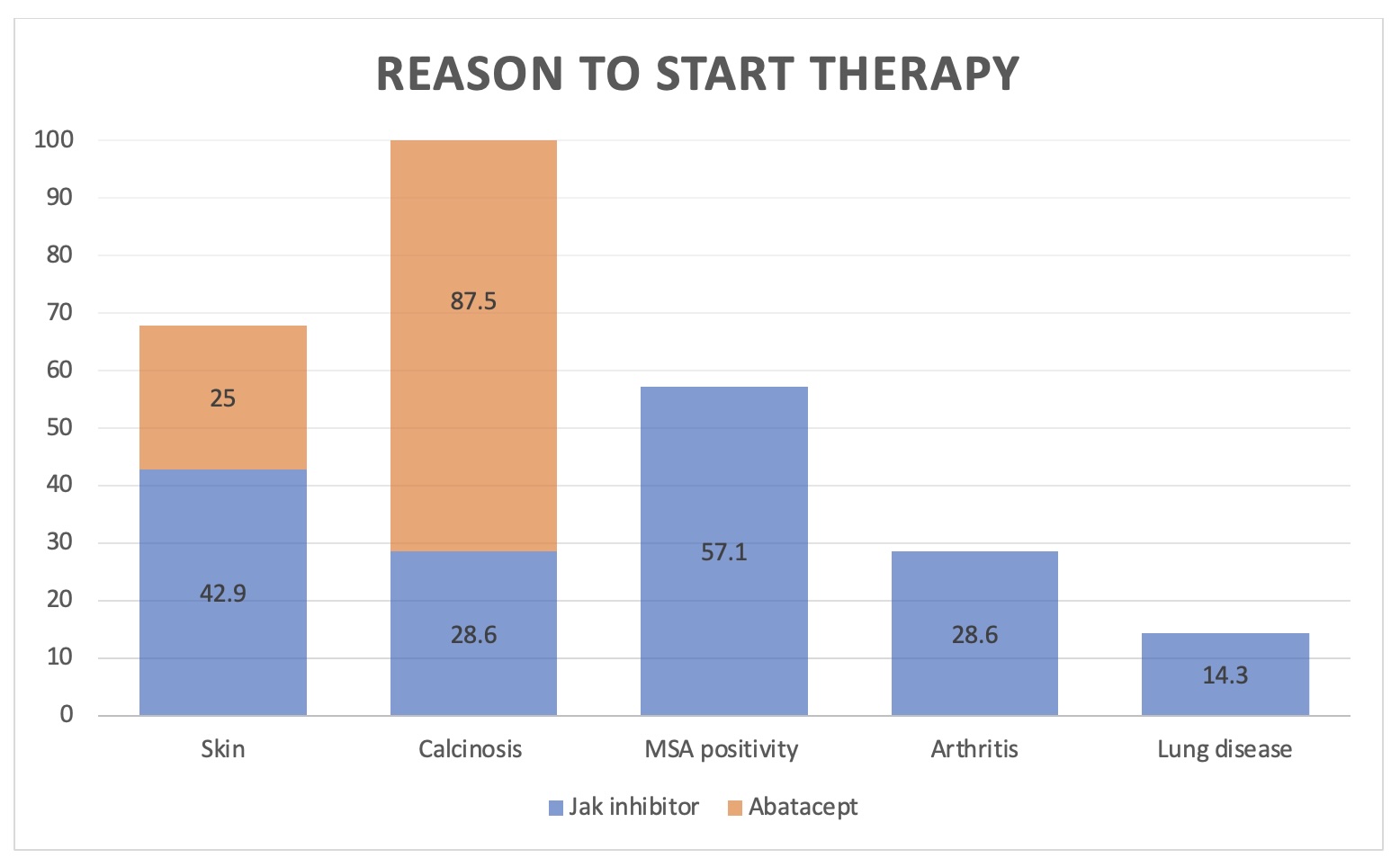
Change in DAS score from month 0 (at diagnosis) and month 12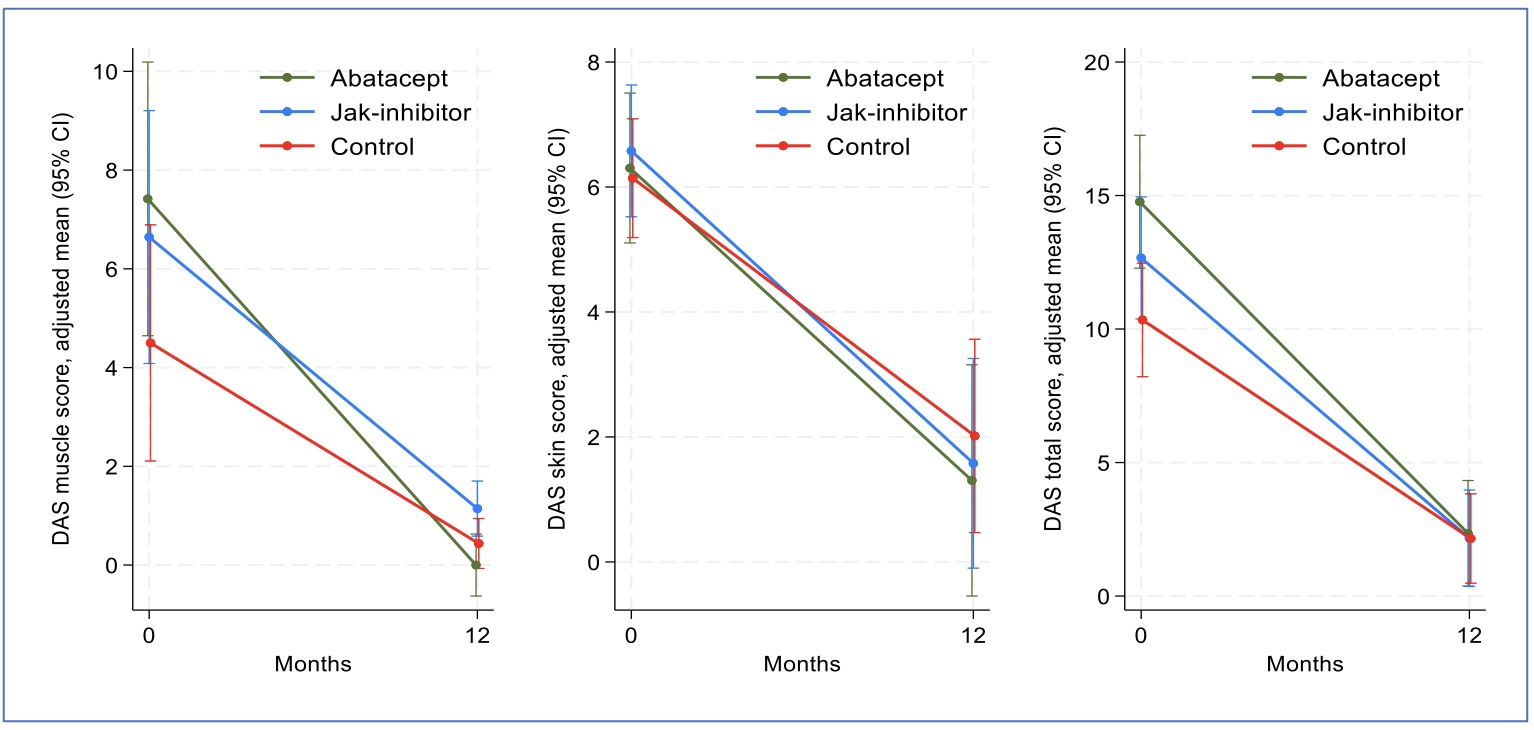
To cite this abstract in AMA style:
Torres Sanchez J, Gist D, DeGuzman M, Aras S, Nguyen D, Nguyen M, Thakral A. Experience with Janus Kinase Inhibitors and Abatacept in Juvenile Dermatomyositis: A Retrospective Study from a Large Tertiary Center [abstract]. Arthritis Rheumatol. 2026; 78 (suppl 3). https://acrabstracts.org/abstract/experience-with-janus-kinase-inhibitors-and-abatacept-in-juvenile-dermatomyositis-a-retrospective-study-from-a-large-tertiary-center/. Accessed .« Back to 2026 Pediatric Rheumatology Symposium
ACR Meeting Abstracts - https://acrabstracts.org/abstract/experience-with-janus-kinase-inhibitors-and-abatacept-in-juvenile-dermatomyositis-a-retrospective-study-from-a-large-tertiary-center/
